Introduction
Most people picture the electron as a tiny ball orbiting the nucleus, like a miniature planet circling a star. That picture was shown to be wrong nearly a century ago — yet it still dominates textbooks and popular explanations. The truth is stranger, and far more interesting. The electron behaves like a wave, not a particle, and the full geometry of that wave cannot even be described in three dimensions. This article explores the evidence that the electron cloud is a 4th-dimensional wave structure, and shows how that single insight resolves the deepest puzzles of quantum mechanics: uncertainty, spin, superposition, and the mysterious jumps between energy levels.
Since J.J. Thomson first discovered the electron in 1897, the assumption has been that the electron acts like a particle. This notion was challenged in the 1920s when it was discovered that electrons produce an interference pattern when fired through two slits — behaviour that is the hallmark of waves, not particles. As the theory of the photon had already suggested that light exhibits a wave-particle duality, the same assumption was applied to matter. This led to the belief that the electron cloud was a probabilistic wave, where the exact location of the particle could never be predicted.
However, this creates serious problems. Up to that point, the scientific method demanded a strict system of determinism. The introduction of probability left a vast hole in our understanding of the atom, and most people still picture electrons circling the atom in concentric rings.
This outdated Bohr model was disproven by the experiments of the 1920s, which divided the investigation of the atom into particle physics and wave mechanics. Yet both branches still hold onto the notion of wave-particle duality, largely due to Einstein's solution to the photoelectric effect and the ultraviolet catastrophe. However, strong evidence exists that both difficulties can be resolved using a purely geometric model employing the musical ratios of the 4th and 5th.
This allows us to reconsider the electron cloud purely from a wave perspective. What emerges is that the electron cloud exhibits a geometric structure that is 4th-dimensional in nature. This explains the quantisation and stability of the electron cloud, and shows how an electron can appear to jump from one shell to the next without passing through the space in between. For a broader treatment of how geometry reshapes our view of wave-particle duality, see the linked article.
Key takeaways
- The electron is not a particle — it is a wave whose geometry requires a 4th dimension to describe properly
- Quantum uncertainty, spin, and superposition all dissolve into logical wave behaviour once the 4D model is adopted
- The 4D geometric model predicts atomic radii for all stable elements within 3 pm — compared to errors of up to 400% in the Bohr model
What is an electron?
Electrons are often considered elementary particles — meaning they cannot be divided into smaller constituent parts, unlike the proton which is formed of quarks. The idea that electrons are tiny particles circling the nucleus is the predominating view held by most people, and even today this model is used to explain the functioning of the atom.
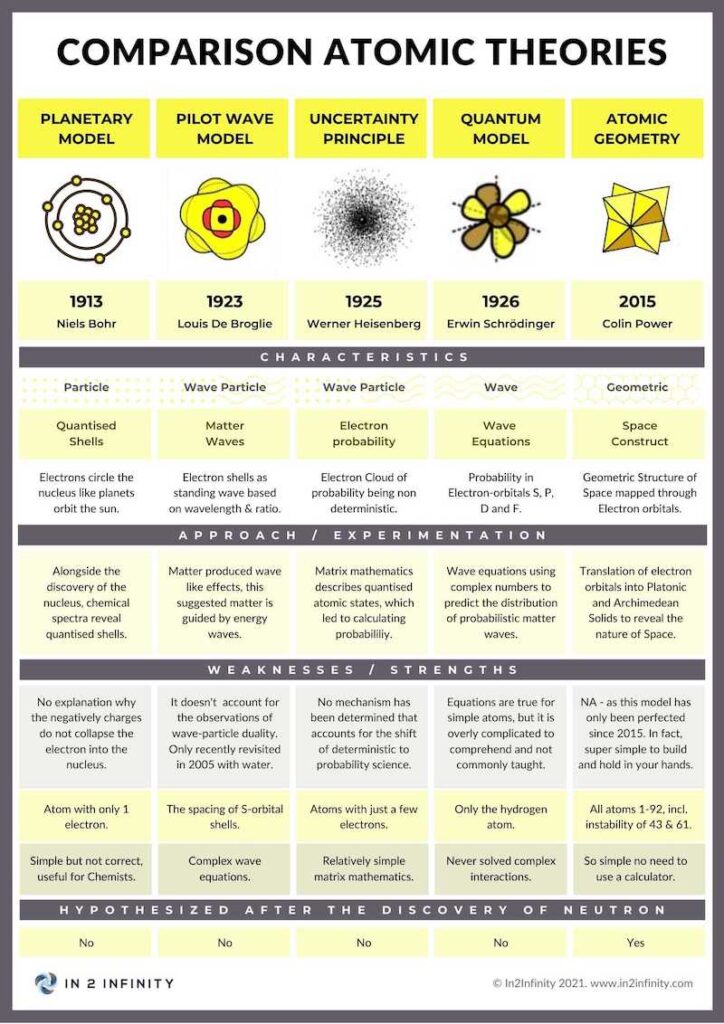
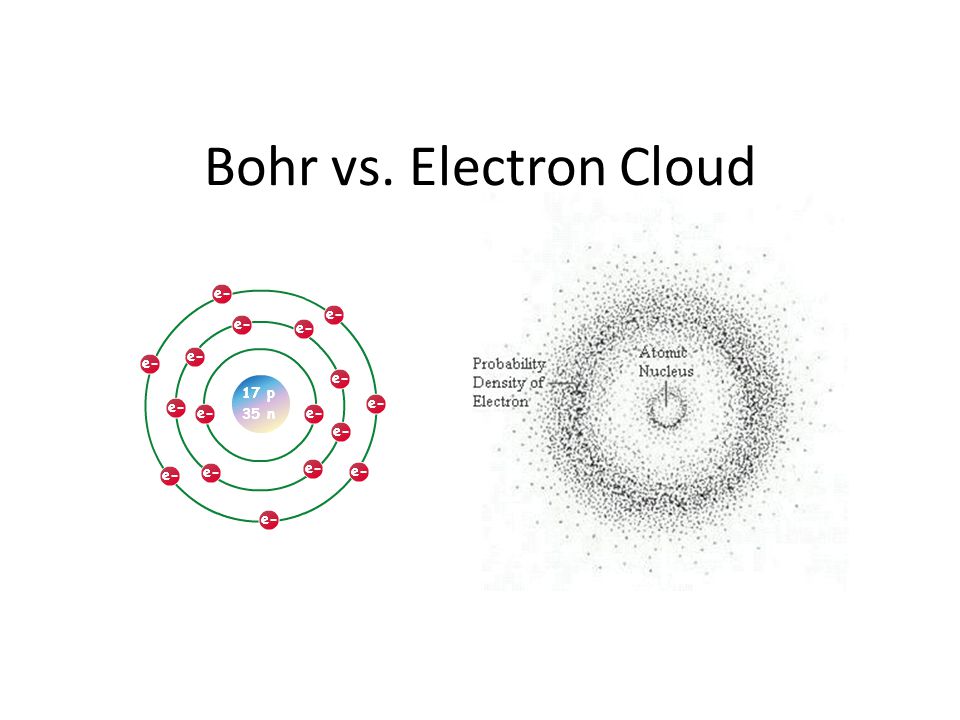
Yet this model was disproven as far back as the 1920s, when the wave nature of the electron was first discovered. When passed through a double slit, electrons produce an interference pattern — the hallmark of a wavelike phenomenon. The same type of experiment was conducted by Thomas Young in 1801, which initially proved that light was a wave. It was only when Albert Einstein suggested the idea of a photon, roughly 100 years later, that the particle model of light resurfaced. This led to the assumption that light exhibits a wave-particle nature. When the wavelike nature of the electron was established, it therefore also adopted the wave-particle duality concept.
However, more often than not, this wave nature is ignored. The main reason for the popularity of the particle model is the simplicity of its explanation. The wave equations proposed by Erwin Schrödinger are so complex that even high-powered computers are unable to solve them. As each electron carries the same amount of charge, the particle model — although incorrect — does allow for a certain level of prediction, which is sufficient to calculate simple molecular bonds. However, the problem remains: the electron is described either as a cloud based on vague notions of probability, or as an oversimplified particle.
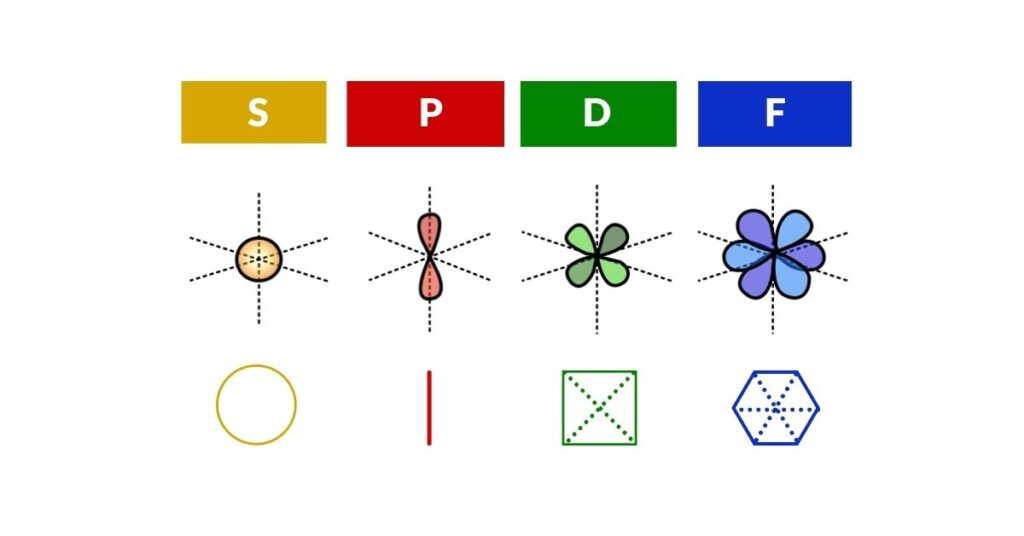
Electron Waves
The wave model of the electron cloud is described by the Schrödinger equations. These complicated mathematical expressions are an expression of a harmonic oscillator — at its simplest, a particle moving back and forth to create a wave. Without going too deep into the mathematics, the result is a series of standing wave models that produce sets of geometric waveforms.
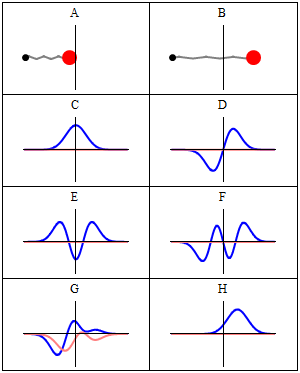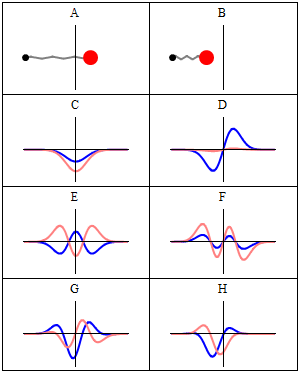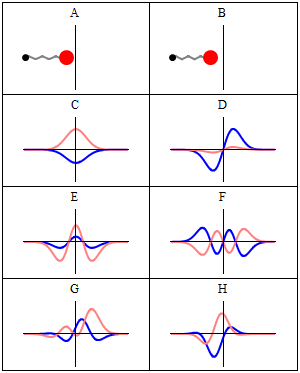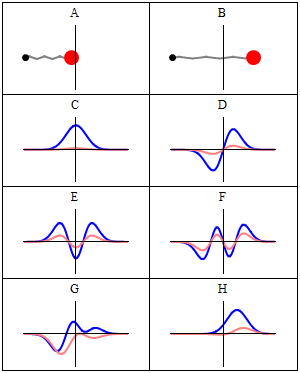
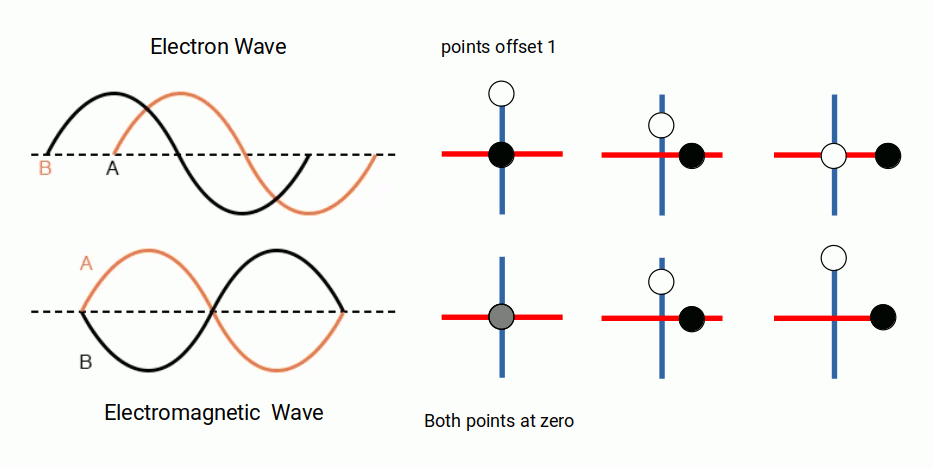
When we consider these waveforms, we cannot help but notice a similarity to the traditional notion of electromagnetic waves, which are also formed of two waveforms offset at 90° to each other — the E and B fields, which are in phase with each other. The electron waves are offset by a quarter of a waveform, meaning there is always an equal distance between the two zero points. It is worth noting that circularly polarised light offsets the E and B fields in the same way as presented in the Schrödinger equations.
The simplicity of this observation is rarely considered. Often the expressions of the Schrödinger waves are superimposed over each other in diagrams, which obscures this curious fact. Whereas previous models of the atom suggested the electron particle orbits the nucleus, the Schrödinger wave model expresses the electron as a standing wave — subsequently verified experimentally by imaging a charged hydrogen atom.
The Electron Charge
Electrons exhibit a negative charge, whereas the proton in the nucleus exhibits a positive charge. The traditional view suggests this is the mechanism by which both particles attract each other. One might expect the electron to plummet towards the nucleus. However, because both particles exhibit exactly the same amount of charge, the electron can only fall as far as the lowest shell — its ground state — where the positive proton and negative electron charges come into balance to produce an overall charge value of zero.
The charge value of the proton and electron is determined by the elementary charge constant (e). In Dimensionless Science, this has a value of ½π. When we consider the offset nature of the electron wave compared to the EM wave, a direct relationship to the unit of elementary charge becomes apparent.
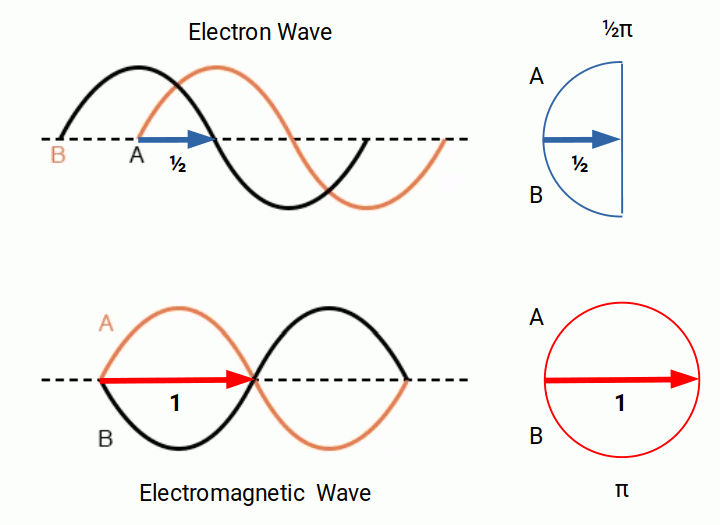
Presently, there is no explanation for why the universe should exhibit charge at all. The wave solution suggests that charge arises from an offset of the waveforms of electromagnetic waves and the wave function of the electron. Electromagnetic waves exhibit a quantum spin value of 1, whereas electrons have a spin value of ½. Whereas an infinite number of EM waves can occupy the same space, only a single electron can occupy a particular position in the electron cloud.
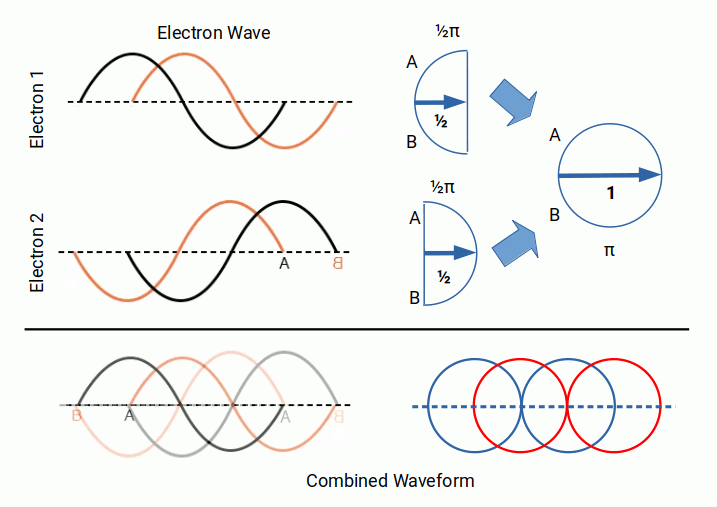
The ½ spin of the electron is the reason why electrons are always found in pairs — one spin-up and one spin-down — with each pair completing an orbital shell. When the unit of elementary charge is multiplied by two, the result is π, and the combined wave comes into the same phase as the electromagnetic wave.
Electron Spin
The concept of electron pairs is expressed by the notion of quantum spin. Each electron exhibits either an up or a down spin orientation. This concept is rather confusing when viewed from the particle model. From the perspective of a wave, however, it simply describes the manner in which the waveform inverts. We have seen how this can unify two waves into phase, bringing the electron cloud into alignment with the EM wave. We also notice that the combined electron wave produces two overlapping circles, as opposed to the EM wave that comprises a single circular wave.
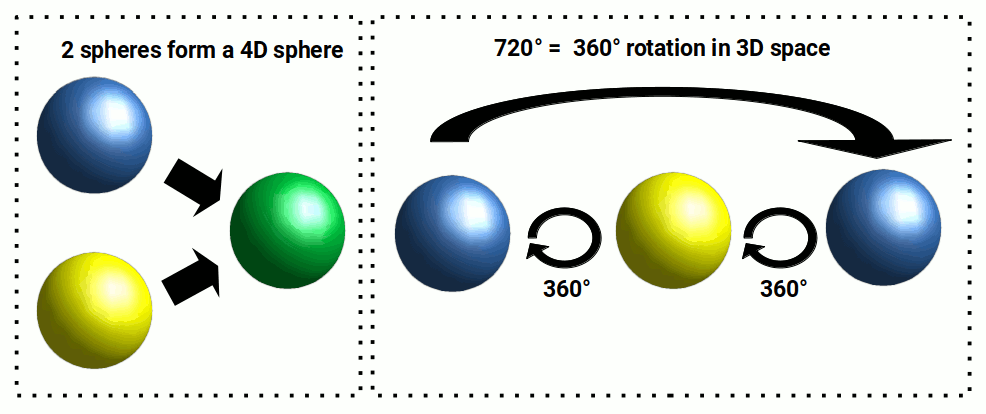
According to quantum field theory, the electron must perform two full rotations in order to complete a single rotation in 3D space. At first, this seems illogical. Yet, once we adopt a 4th-dimensional view of the electron cloud, the notion begins to make perfect sense.
A 4D sphere appears in 3D as two spheres superimposed over each other. When it rotates 360° through its time axis (W-axis), the first sphere swaps places with the second. Upon the next 360° rotation, the spheres swap back, completing a full cycle. In total, the 4D sphere has rotated 720° to return to its original starting point.
The concepts of 4D space are still relatively unfamiliar, but they become much clearer with the example of a hypercube. Unlike the sphere, the rotation of a cube produces a shadow projection that changes visibly as it moves.

The torus is another 4D object whose field behaves in exactly the same way and can produce the ½ spin of the electron cloud. Compared to light — which is an expanding sphere — the torus acts as a container for electron energy. This fundamental difference between electromagnetic energy and the energy of the electron is verified by the behaviour of signal transmission. When an antenna is charged, it forms a near field shaped like a torus. Once the signal passes beyond this near field, it expands to infinity at the speed of light. All matter exhibits such a near field at its surface boundary, known as evanescent waves, generated by the electron cloud. This is why we do not actually touch anything — we only experience the repulsion of the electron field.
Uncertainty Principle
One of the key claims of quantum mechanics is that a particle can be in multiple places at the same time, yet when observed or measured it collapses into a single state. From the perspective of the wave model, the resolution is straightforward.
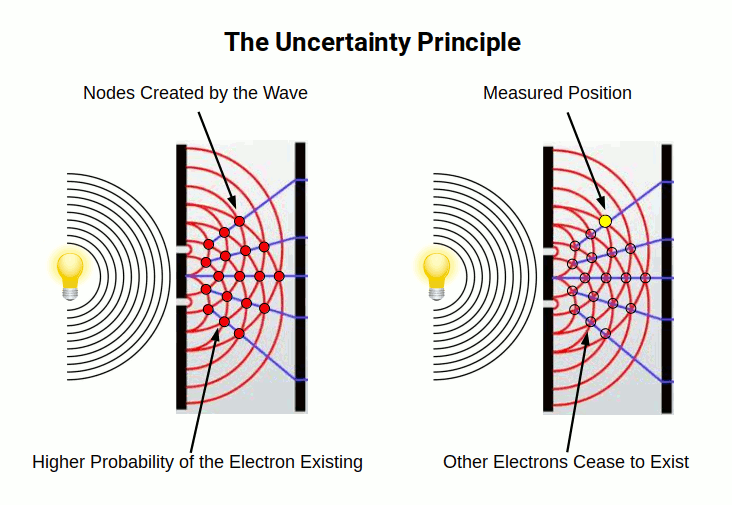
When an electron beam passes through a double slit, it forms an interference pattern. The two interfering waves create overlapping nodes which amplify the wave at specific points. Where the peak of one wave meets the trough of another, the waves cancel out. This constructive and destructive interference is characteristic of all wavelike phenomena.

The difficulty arises when we try to describe this wave behaviour from the perspective of a particle. A measurement at a particular location of the wave — where the amplitude peaks — is interpreted as the electron having been "found" there. The problem with the particle model is the assumption that once the electron has been located, it ceases to exist everywhere else. This is simply not true. The electron wave continues to exist at all the points where the wave peaks. It does not collapse into a single location, as quantum mechanics suggests.
The Uncertainty Principle was first proposed by Werner Heisenberg in 1927, shortly after the wavelike nature of the electron — suggested by Louis de Broglie in 1924 — was verified by the Davisson–Germer experiment. In order to salvage the failing Bohr model, the concepts of uncertainty and probability were introduced. This became the Copenhagen interpretation, which remains the prevailing model of particle physics. At around the same time, the Schrödinger equations were formulated, offering a far more accurate prediction of electron behaviour based on wave interference. The introduction of probability was vigorously opposed by many scientists of the time, including Albert Einstein.
The Schrödinger's Cat thought experiment was originally proposed by Erwin Schrödinger to highlight the absurdity of applying probability to macroscopic reality. Ironically, it was subsequently used to promote the supposed "weirdness" of quantum physics. The Observer Effect extended the Uncertainty Principle further, suggesting that reality only exists once it is observed. We challenge this view with the theory of the 4D Aether, which dispenses with wave-particle duality and maintains that the moon exists whether or not anyone is looking at it.

Video animations that seem to demonstrate wave-particle duality often depict the idea that placing a detector at the slit causes the interference pattern to disappear, replaced by two straight lines indicative of a particle. This has never actually been observed — it was only a thought experiment by John Wheeler. The detection of an apparent single particle is produced by filtering the light or electron source until only a single point is detected. In other words, it is the threshold of detection that creates the illusion of a particle. Over time, the interference pattern always re-emerges.
The probabilistic nature of the electron, and all the theories that emanate from it — the Observer Effect, Many Worlds Theory, Bell's inequality — can be resolved simply by viewing the electron as a wave. The wave-only view is made possible once the wave solutions to the photoelectric effect and the ultraviolet catastrophe remove the need for the photon particle. This returns scientific enquiry to logical coherence, rather than thought experiments with no foundation in physical reality.
Probability Waves
To appreciate how much better the 4D model performs, it helps to first see just how badly conventional quantum models fail at predicting something as basic as atomic size — even for the simplest element in existence.
When the wavelike nature of the electron became apparent, probability theory was introduced to describe it. By taking multiple point measurements, a probability curve is generated, representing the likelihood of finding the electron at any given point.
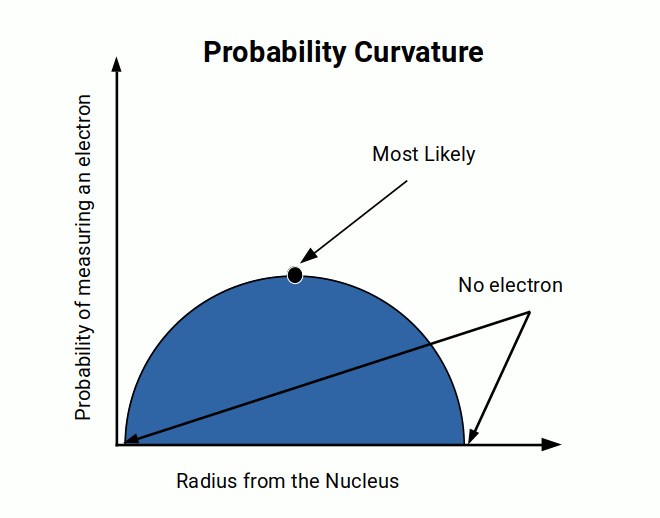
The space inside the curvature is termed the electron density. The wave model resolves this simply by distributing the electron cloud over an area surrounding the nucleus — no probabilistic description is needed once we discard the assumption that the electron is a particle.
One of the most striking problems with modern quantum theory is this: while it claims to predict atomic behaviour to extraordinary precision, it fails to come close to the experimentally observed radius of even the simplest atom, hydrogen. According to the Bohr radius, the radius of the hydrogen atom is around 53 picometres. Yet experimental measurements give a radius of only 25 picometres — an error of over 100%. For the second element, helium, the Bohr model predicts 31 pm while experiments give 120 pm, a disparity of nearly 400%.
To manage these shortcomings, a second set of measurements was derived — the Van der Waals radius — which treats the atom as a hard shell at the electron's furthest possible point from the nucleus. This gives 140 pm for helium, only 20 pm off. Because each atom's radius varies widely across the periodic table, chemists must swap between the two systems to preserve the mathematical integrity of calculations.
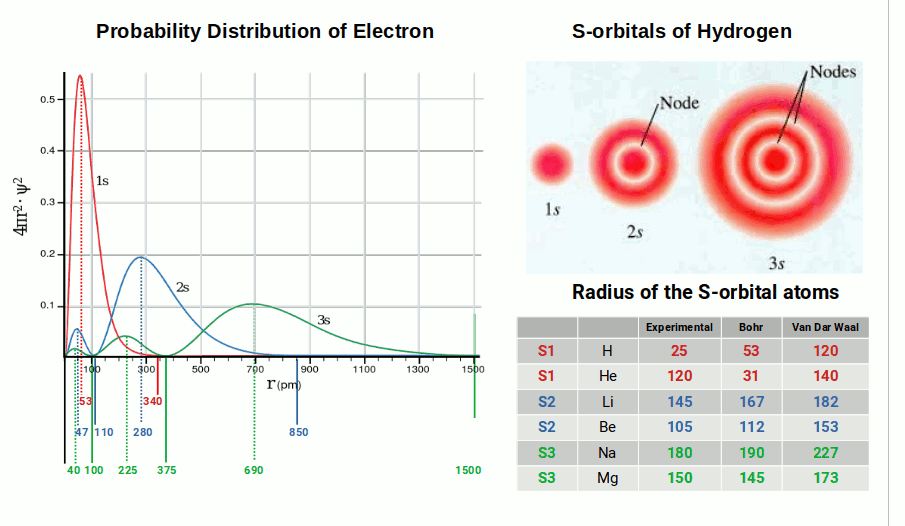
The mathematics of electron density therefore employs various systems to produce accurate predictions — a problem that persists to this day and has been noted by chemists who struggle to find a quantum model that fits observed phenomena.
As scientific exploration of the atom advances, the need for accurate predictions becomes ever more important. However, once we adopt a geometric view of the electron cloud, something remarkable happens: it becomes possible to produce a single model that predicts the atomic radii of all stable atoms within a 3 picometre margin of error.
The theory of Geoquantum Mechanics is the first model of the atom that considers both the nucleus and electron cloud from the perspective of 4D geometry. It accurately explains the reason for lanthanide contraction and the instability of elements 43 and 61 on the periodic table. This geometric approach has deep connections to the broader framework of Atomic Geometry.

Superposition
The quantum mechanical concept of superposition spans a variety of phenomena and is one of the most misunderstood concepts of modern science. In essence, it suggests that a particle can exist in multiple places at the same time. As with the Uncertainty Principle, much of this can be described simply once we adopt a wave view of the electron cloud. The wave spreads over an area, and readings can be taken from different points in space to locate the supposed particle. However, developments in wave-particle theory have evolved more elaborate definitions.
One key example comes from the Stern–Gerlach experiment, first conducted in 1922. Silver atoms were evaporated at high temperatures and passed through a magnetic field that deflects their trajectory. If the electron were a small particle circling the atom, the deflection would vary continuously with the electron's position. Instead, the atoms are deflected either up or down, forming two distinct lines on a photographic plate. This experiment was one of the first clear demonstrations of electron spin. It is not that the electron particle is spinning — it is that the electron field has a north and south, or up and down, polarity, just like a torus field. The atom is deflected up or down depending on its orientation.
When electron spin is viewed from the perspective of a 4D torus, the up and down states are simply the expression of the north and south poles swapping as the 4D object rotates through time. Just as with the hypercube — which exhibits only two possible states, with each cube falling into alignment with the other — the north and south poles alternate. As the torus passes through the magnetic field, each atom is oriented either pole-up or pole-down, causing deflection in two discrete directions.
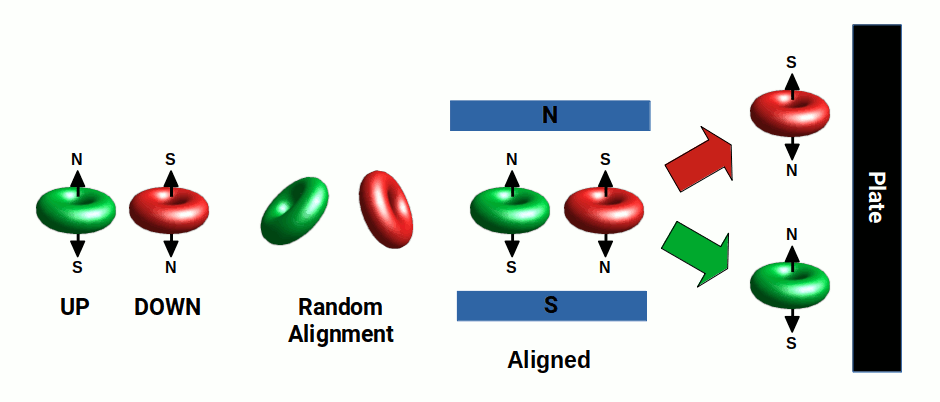
Variations of the Stern–Gerlach experiment involve placing more than one magnetic field in series, with one pathway blocked. When both fields are oriented in the same direction, the beam continues to be deflected in the same orientation. When the second magnetic field is rotated 90°, the beam is again divided into two parts. This is the expected outcome of a 4D wave with ½ spin. Even electrons selected for an up-spin can be equally divided into +x and -x directions by a 90° field, indicating that spin is not confined to a single axis — each electron contains both up/down and left/right qualities. This, too, is readily explained by the 4D torus field.

This simple notion of quantum superposition has been misrepresented in some quantum physics teaching, most notably in the use of "colour" and "hardness" as labels for the x and z magnetic orientations. This inaccurate framing — addressing the Stern–Gerlach results from a particle-only perspective — leads to irrational conclusions such as the electron acting in multiple states simultaneously while unobserved. Once we identify the wave nature of the electron, superposition returns to the logical domain of waves in specific phase orientations.
Quantisation of the Electron Cloud
Another form of quantum superposition arises from the nature of the electron cloud surrounding the atom. When a light wave of a certain frequency strikes an atom, the electron cloud becomes excited and increases in size — traditionally described as the electron "jumping" to a higher energy shell. In fact, when excited, the atom fluctuates between two states.
The claim that the electron occupies two states simultaneously until observed is impossible to prove, since the act of observation is destructive to the atom. The atom alternates between excited and non-excited states in a series; any measurement captures only the state at that particular moment. Just as superposition in electron beams is resolved by the wave model, so too is the behaviour of the electron cloud. This principle lies at the heart of quantum computing.
There is, however, an important distinction. The electron cloud does not gradually expand — it jumps discretely, and no measurement will ever detect an intermediate state. This is why quantised reality is so widely accepted. The wave model explains it equally well: the electron can only exist where the shells fall into a specific resonance, which is why only light at a specific frequency can excite the cloud to a higher level.
From the perspective of a 4D electron cloud, this quantisation is natural. A 4D object does not exist entirely within 3D space. When it rotates through its time (W) axis, it moves between two states. The absorption of an EM wave triggers this rotation, which expands the form in 4D space so that it appears — apparently instantaneously — in the higher shell in 3D space.
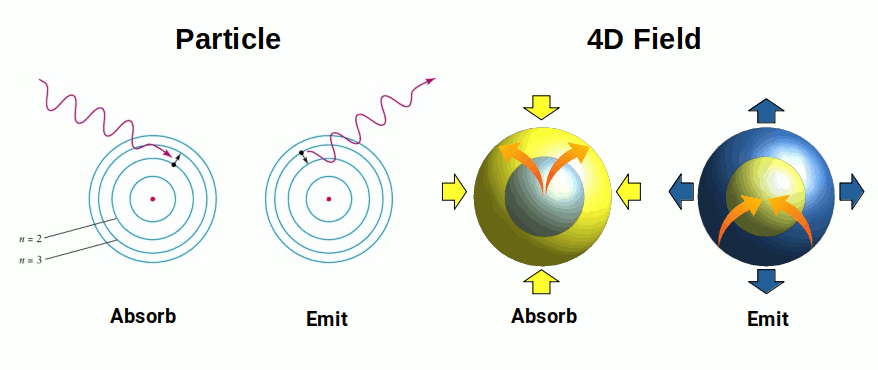
In the particle view, a single electron absorbs a photon, which makes it jump to a higher shell; when it falls back, a single photon is emitted. In reality, light approaches an atom from all directions simultaneously, with many photons potentially striking the atom at once. A linear particle trajectory cannot account for this. In the 4D wave view, light is absorbed and emitted in all directions, maintaining the spherical nature of the light wave. For more detail on the geometric structure of the electron cloud and how this framework relates to the nature of light itself, see our treatment of why light does not have mass and the full framework of Atomic Geometry.
Conclusion
The 4D electron cloud model proposes that the electron exists as a wave whose quantisation arises from the 4th-dimensional nature of space. This explains why there are only two possible spin states — up and down — as the rotation of the 4D sphere swaps the magnetic poles, producing two distinct lines on an imaging plate when atoms pass through a magnetic field. The model removes any need for probability, since the wave is distributed over an area surrounding the nucleus — explaining why the electron radius has never been pinned to a single point. The quantisation of the electron into discrete energy bands, and the requirement that only specific frequencies of light can excite it to a higher level, both follow naturally from 4D geometry.
The traditional particle model has dominated quantum science because it is simpler to teach and sufficient for basic chemistry. However, its failure to predict accurate atomic radii — with errors of up to 400% for common elements — reveals its fundamental inadequacy. The 4D geometric wave model, developed within the framework of Geoquantum Mechanics and the wider theory of Atomic Geometry, reproduces experimentally measured radii for all stable elements and offers a coherent, deterministic alternative to a century of quantum ambiguity. If you are interested in how related phenomena such as the cosmic microwave background fit within a geometric model of the universe, those connections are explored in the linked articles.
FAQ
Surely if the electron was a wave, someone would have suggested this before?
You might have thought so, however, as the particle nature of light was already established, the assumption has always been that the electron is a particle. When the search for the Aether was abandoned, the particle nature was required to explain how light and electrons could travel through the vacuum of space. Ironically, the idea of the quantum field had to be reintroduced, but the particle model still prevailed as the idea of probability became adopted.
I thought the electron has a mass, so it must be a particle?
In the standard model, there are also massless particles, such as the photon. However, the mass of an electron was first calculated by J.J. Thomson by firing an electron beam through an electromagnetic field and measuring the deflection. The mass also changes dependent on velocity, so the electron mass is calculated when not at rest. No one has ever weighed an electron directly.
What does the 4D electron cloud model mean for quantum theory?
The traditional particle interaction view dominates quantum science, but it fails to predict more complex systems such as those in quantum chemistry. The 4D geometric wave model reproduces experimentally measured radii for all stable elements, whereas the Bohr and Van der Waals radius models show inaccuracies of up to 400%.
Why does the electron cloud appear quantised in discrete energy levels?
From the perspective of a 4D electron cloud, quantisation is straightforward. A 4D object does not exist completely within 3D space. When it rotates through its time (W) axis, it moves between two states. The absorption of an EM wave of a specific frequency triggers this rotation, which causes the cloud to expand in 4D space and appear in a higher shell in 3D space — with no detectable in-between state.




