Introduction
Most people think of the electron as a tiny particle that orbits the atomic nucleus. In truth, there are four types of orbital — S, P, D, and F — each of which creates a different geometric pattern. In this article we examine the 2D orbital geometries of the electron cloud and offer a simple geometric explanation for the different configurations.
The core argument is straightforward: the four orbital types correspond exactly to the only four geometric primitives that can be constructed using the two types of regular 2D tessellation — dot, line, cross, and hexagon. This geometric constraint explains why there are precisely four orbital types and no more. By extending the analysis into 4D geometry, we can also explain why electron spin has exactly two quantised values.
For the geometric treatment of individual orbital shapes, see S-orbital geometry, P-orbital geometry, and D-orbital geometry.
Key Takeaways
- The four orbital types (S, P, D, F) correspond exactly to the only four geometric primitives constructible from regular 2D tessellation: dot, line, cross, and hexagon — explaining why there are precisely four orbital types and no more.
- Electron spin has exactly two quantised values because electrons can only appear with up or down orientation, matching the fundamental limitation of two-colour tessellations: after the hexagon, no further two-colour regular tessellation is possible.
- The entire orbital sequence can be derived from pure geometry without equations: a sphere divides into two creating a line, the line doubles and rotates 90° creating a cross, and additional spacing at 60° creates a hexagon.
What is an orbital?
Traditionally, an electron orbital is considered to be a probabilistic region of space that determines the likelihood of finding an electron at a particular point around the nucleus. Each orbital represents a specific energy level of the electron. When an electromagnetic wave strikes the atom, certain frequencies are absorbed and the electron jumps into a higher orbital shell. The atom becomes energised. Subsequently the electron falls back down into the original shell and a new electromagnetic wave is emitted.
This can be considered as the vibration of an atom, but with one key difference. The electron does not move through space — it literally jumps. This means there are certain areas surrounding the atom where the electron will never be found. This fundamental nature of quantisation means the energy levels are divided into discrete steps, similar to a piano keyboard: each note produces a particular frequency tuned to all the other notes, and as we move from one note to the next there are discrete jumps based on the original root note.
Orbital geometry and the 2D plane
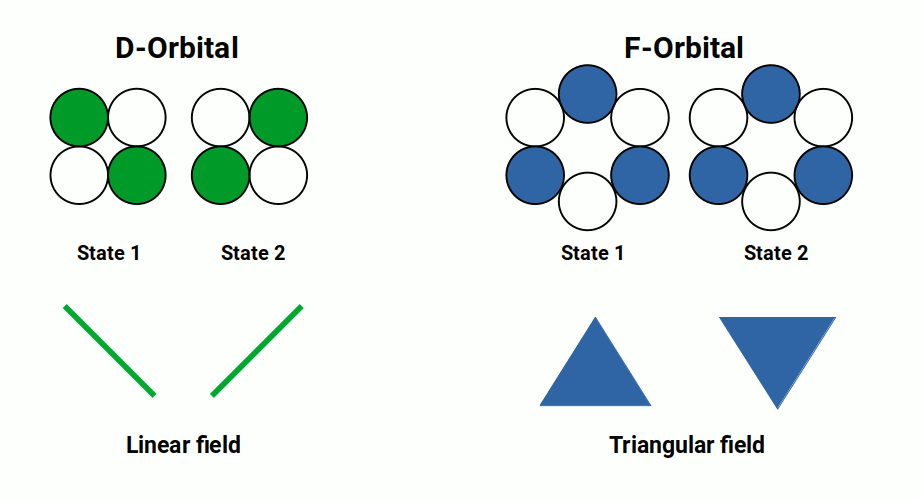
Based on geometric principles, the orbital shapes can be described precisely without complicated equations. The first orbital type appears as a simple sphere — represented as a single dot. The next orbital to emerge is the P-orbital, where the sphere doubles to become two spheres; when the dot divides into two, a line is created. In 2D Euclidean geometry a line is always the shortest distance between two points. The D-orbitals form next, whereby the number of dots doubles again to create a new line rotated 90° to the first — connecting the ends of these lines produces a square (cross). The final F-orbital type adds another line; these lines become evenly spaced at 60° intervals and joining their ends creates a hexagon.
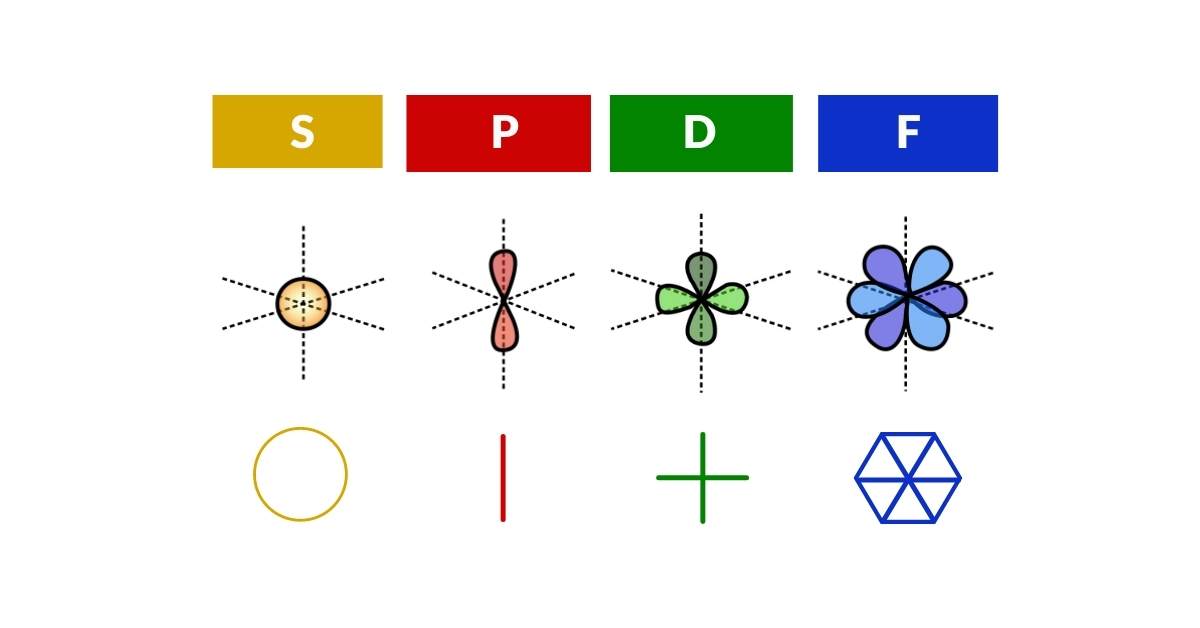
No orbital geometries have ever been established beyond the F-orbital type. Theoretical G-orbitals are sometimes considered but have never been observed. Geometry provides a simple explanation: there are only two types of regular 2D tessellation constructible using two colours. The most familiar is the square tessellation — the chequered plane of a chess board. The second is the triangular tessellation, where six triangles group to form a hexagon. Crucially, the hexagonal tessellation requires three colours to tile the 2D plane. Since electrons can only appear with either up or down spin, the limitation to exactly two states is perfectly expressed by these two types of regular 2D space. After the hexagon, no new two-colour tessellation is possible — and so the F-orbital is the last.
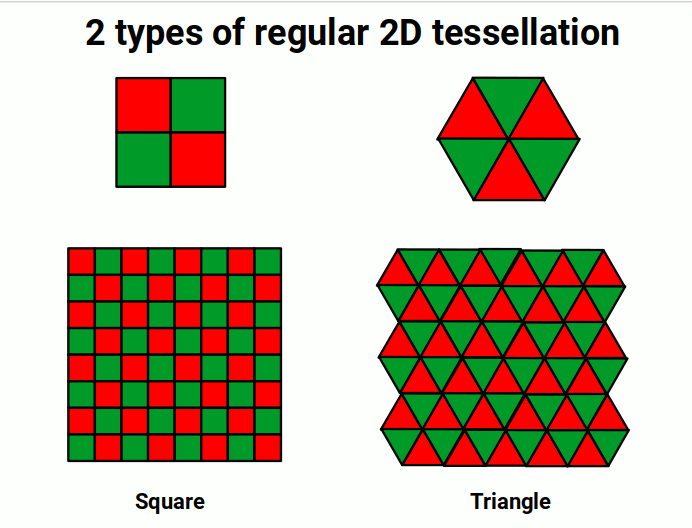
At first glance it appears that each orbital follows a doubling pattern: from a dot that divides into two to form a line, then a line that divides again to form a cross. One might expect the next division to produce a double cross (octagon). However, in the transition from the D to the F-orbital, this doubling sequence does not continue — the F-orbital falls onto the triangular tessellation instead. After this, the pattern terminates. The principles of 2D geometry therefore define both the limitation of orbital shapes and the sequence of their evolution.
This concept is simple enough to raise the question of why it has not been considered before. In standard mathematical thinking it is often held that the second dimension can only exist when embodied in the third dimension — this view begins to shift that belief. In physics, results are typically determined through the experimental measurement of a single point in space; the measurement problem in quantum mechanics is derived from this very nature. The assumption that the electron is a particle, combined with the adoption of wave-particle duality, has steered analysis away from the geometric perspective.
Electron orbitals and compass construction
The geometric sequence described above — dot, line, cross, hexagon — emerges naturally from the ancient art of compass construction. Compass construction explores geometry through pure proportion, producing designs without measurement. This matters because the universe itself does not use mathematics to calculate emergent phenomena; everything is relative to the smallest scale. In science this has been quantified as the Planck length, derived from the Planck constant, which defines the minimum distance at which a measurement of physical reality can be ascertained.
Planck Length = √ ħG/c³
Where ħ is the reduced Planck constant, G is the gravitational constant and c is the speed of light.
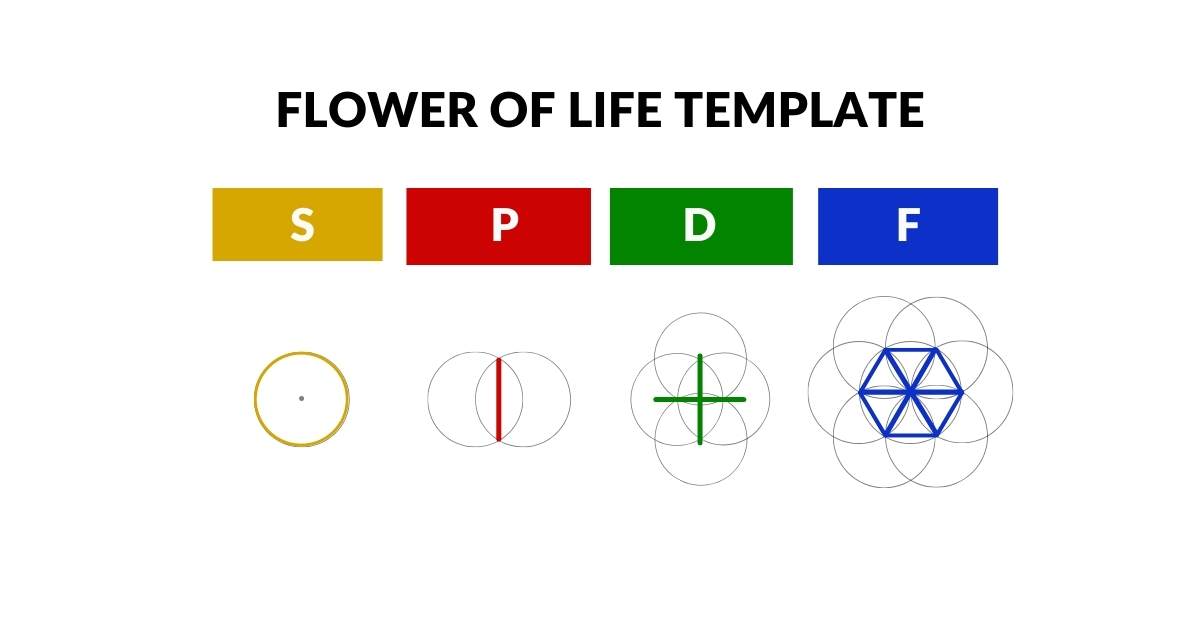
When a compass is used to inscribe overlapping circles of equal radius, a pattern known as the Seed of Life naturally produces the dot, line, cross, and hexagon in sequence — the same sequence followed by S, P, D, and F-orbitals. The pattern does not continue beyond this point: the geometry terminates at the same boundary that the orbital types do.
The 4D electron
Whilst the correlation with 2D geometry perfectly describes the evolution of orbital types, it does not by itself resolve the curious nature of electron spin. To explain that, we need to examine 4D geometry — a prospect that is less daunting than it sounds.
4D geometry is simply an extension one dimension above 3D. A 1D line with a length of 2 squares to give 4 — the side of a square comprised of 4 smaller squares. Raising 2 to the power of 3 gives 8 — the number of cubes in a 3D cube. Raising 2 to the power of 4 gives 16 — the number of smaller cubes in a 4D hypercube. Each time we raise to a higher power, the result defines the next dimension.
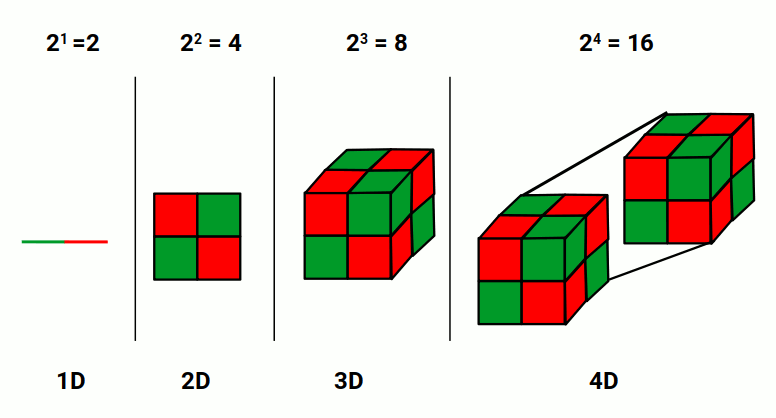
The 4D hypercube is essentially two 3D cubes that occupy exactly the same location in 3D space. Because we cannot reproduce a 4D object in 3D space, shadow projection is used to explore higher-dimensional forms. Each 4D shape has x, y, and z spatial dimensions plus a W dimension. When the 4D hypercube is rotated on the W-axis, the two cubes swap places.
A useful analogy is a flipbook: each page is a flat 2D image, but flipping through the pages creates the impression of continuous motion. What we perceive as movement through time is actually a sequence of discrete 3D frames. In the same way, the 4D rotation of the electron field presents two discrete states — up spin and down spin — in sequence, with no intermediate position. The electron does not transition smoothly between states any more than a flipbook frame can be "between" two pages.
The 4D hypercube volume can also be calculated by multiplying two 2D planes: 2² × 2² = 16, or 2⁴. This means two intersecting 2D planes of opposite colour orientation — one in the manifest 3D space, one in 4D space — swap places in quantised steps just like the electron that jumps location without moving through space. The colours swap discretely, corresponding directly to up and down spin.
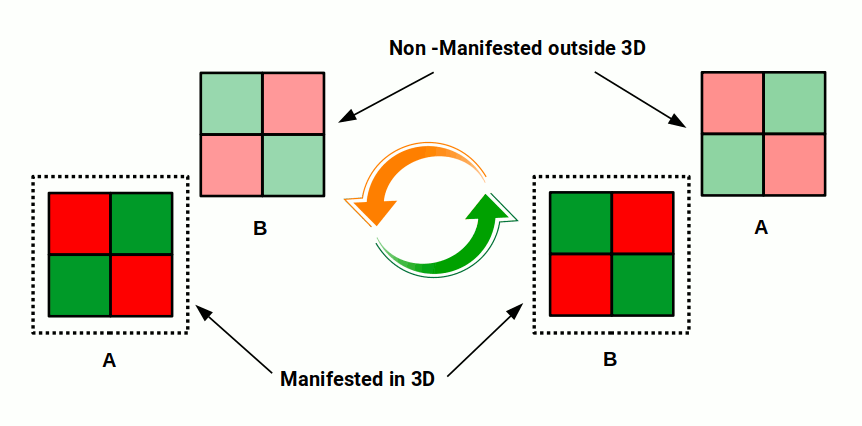
This model is the only one of its kind that properly explains the quantised nature of electron spin. Unlike the classical wave model, at no point will the electron ever be found in the zero position. The quantisation of reality is explained by the rotation of a 4D object in 3D space.
This is based on the Euclidean notion of 4D, which differs from the conventional 4D spaces of Minkowski spacetime. The key difference is that this model shows how time itself can be quantised into discrete units — moments of time are rendered in 3D space sequentially. The two cube orientations of the hypercube are inverse to each other: the first shows an electron with up spin, the second with down spin.

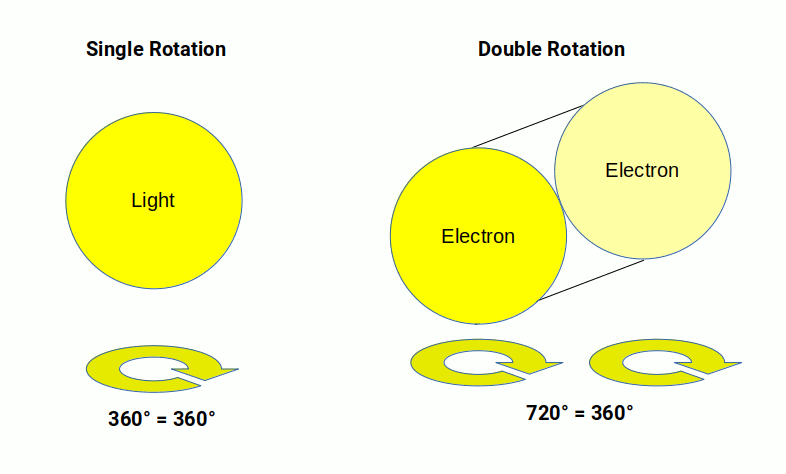
This radically redefines the notion of time. It explains why time appears to have only a forwards motion, accounts for the quantised up and down electron spin states, and is coherent with mathematical theories of quantum spin through spinors, which require a 720° rotation to complete a single rotation in 3D space.
The wave function requires ψ to be squared for the system to work, removing the minus sign from the asymmetric wave function — just as we have squared the two types of 2D plane. The mathematical notion that negative square numbers produce a positive result is challenged in the theory of Geometric Maths, which re-examines the foundational principles of number theory and is able to resolve the Continuum Hypothesis through the perspective of the 4th dimension.
In quantum theory the spinor must rotate 720° in 3D space to complete a single rotation, yet the electron cannot physically be rotating as that would require superluminal speed. The 4D perspective resolves this without compromising a logical notion of spacetime. Rather than seeing the electron as a particle, the model proposes that the electron cloud is a 4D field.
Electron particle vs field
The question of whether the electron is a particle or a field has significant consequences for how we model the orbital structure. Standard quantum mechanics treats the electron as a particle and describes its location probabilistically — but this approach cannot explain the quantised nature of spin from first principles.
From a 4D geometrical perspective, both the static and fluid aspects of the electron field are satisfied simultaneously. All atoms are immersed in a quantum foam — the foundational source of all energy in the universe, variously called vacuum energy, cosmic microwave background, or the quantum field depending on the discipline, but with the same core premise. The 4D Aether provides a detailed wave-only treatment of the photoelectric effect and the ultraviolet catastrophe within this framework.
Once this field perspective is adopted, simple explanations for the orbital types begin to emerge. A single disturbance on the surface of water creates concentric rings that expand as a circular wave. In 3D space this becomes a spherical wave, accurately describing the nature of light and explaining why light intensity diminishes by the square of distance (the inverse square law).
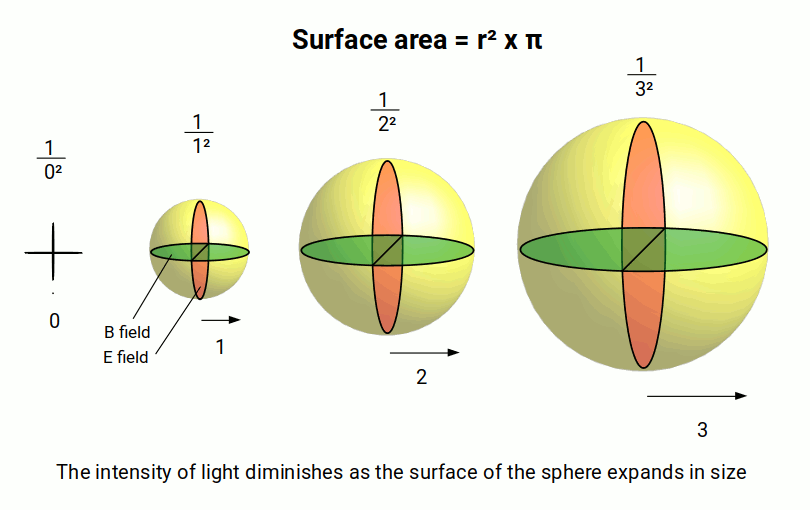
For the electron field, the first orbital geometry is also a sphere. However, this field is contained within the radius of the atom rather than expanding outward. In standard theory this is due to the quantised spin states of the electron, which require a 720° rotation in 3D space — a property inherent to all fermion particles. Light, by contrast, acts as a normal vector with a 360° rotation in 1:1 ratio with 3D space, making it a boson. This key difference defines the electron shells and prevents the electron field from collapsing.
The double rotation of the electron acts as a container for the vacuum energy, increasing the energy density inside the field compared to outside. This explains why only certain frequencies of light are absorbed by the electron cloud. A particular frequency is completely absorbed, causing the electron to jump to a higher energy shell. In 4D terms this motion is triggered by that frequency producing a single rotation of the 4D field — the field rotates outside 3D reality and reappears at a higher energy level, before falling back and emitting a new wave on the second rotation. The process is governed by the 4D rotation ratio between the light wave and the electron cloud, consistent with the fact that the speed of light is limited by the electromagnetic resistance of the vacuum.
S and P orbital geometry
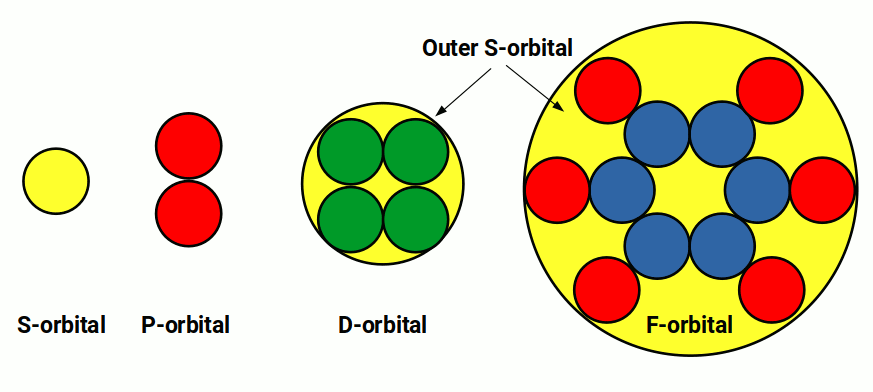
The S, P, D, and F orbitals appear in a specific order. The first shell of the electron cloud is completed by Hydrogen (1) and Helium (2), which comprise a single S-orbital. In terms of 4D geometry this forms a 4D sphere where each electron exhibits an up or down spin. Helium (2) is a noble gas — its electrons are fixed and cannot be removed to form molecules. This completes the first shell upon which all subsequent elements are built.
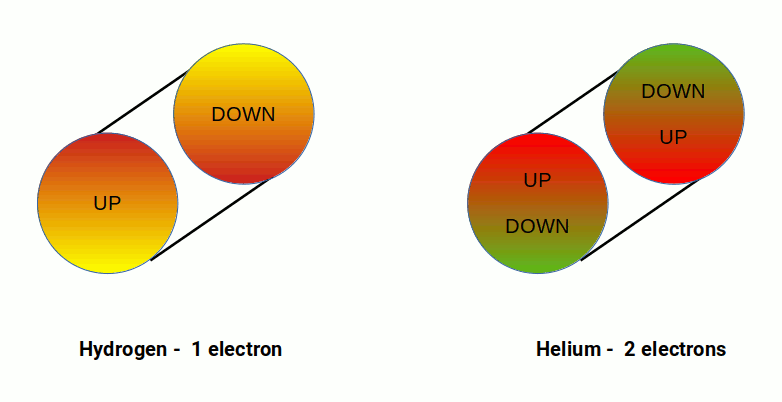
When the electron is viewed as a 4D field, this boundary acts as a container for the vacuum energy. When the up and down spin states are both filled, the 4D sphere reaches magnetic equilibrium, processing both states simultaneously. This explains why atoms with two electrons in their outer S-orbital shell are not deflected in the Stern-Gerlach experiment.
In the 2nd shell, another S-orbital forms for Lithium (3) and Beryllium (4) — a double hypersphere. Unlike Helium, Beryllium does not form a noble gas; its outer two electrons can bond with other atoms while the inner shell retains its inert qualities.
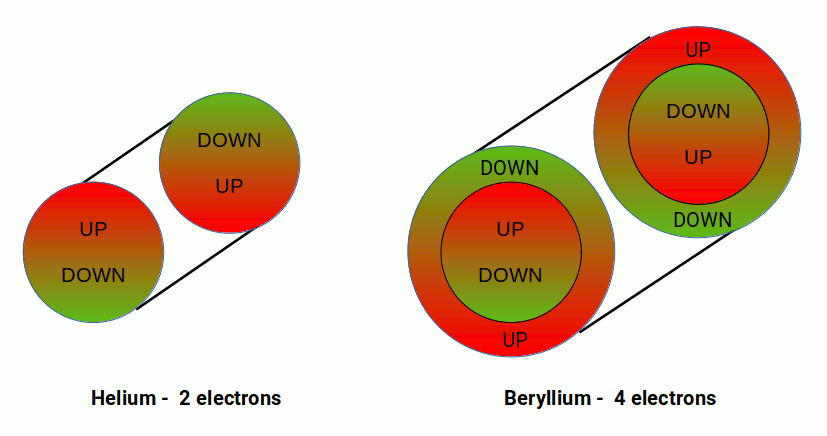
After this, the next six elements fill three P-orbitals. The first three electrons fill one side of the set before the remaining ones complete the opposite lobe — this is the Pauli exclusion principle.
P-orbitals appear outside the S-orbital sphere, creating an extended lobe further from the nucleus that is more likely to form bonds with other atoms. This geometric extension is the basis of molecular geometry. As P-orbitals come in sets of three, they can be mapped onto the Flower of Life mandala — a simple extension of the Seed of Life.
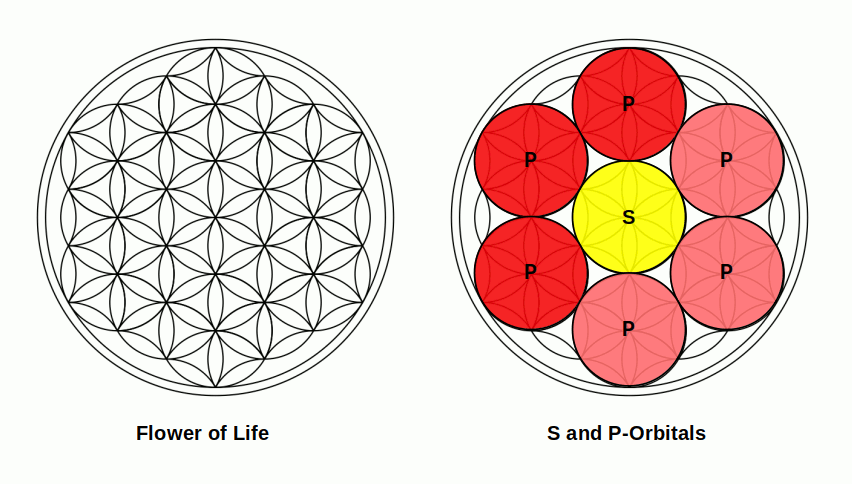
Once the P-orbital set completes, the second noble gas Neon (10) forms. Like Helium, none of Neon's electrons can form atomic bonds, creating a second closed boundary.
Both S and P-orbitals share a geometric commonality: they are not derived from the 2D plane but from a dot that divides to create a line. From the perspective of 4D, both are types of 4D sphere. S-orbitals are a 4D sphere superimposed in the same space; P-orbitals are a 4D torus — geometrically akin to the sphere, and indeed P-orbitals are sometimes visualised as a torus field rather than two spherical lobes at opposite sides of the nucleus.
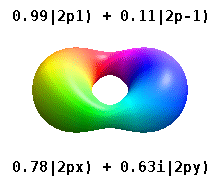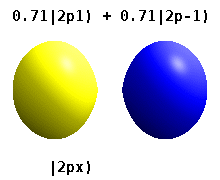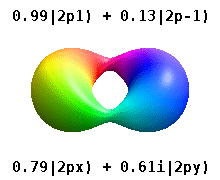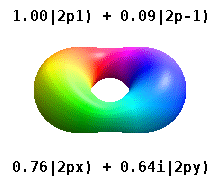
Aside from Helium, all non-reactive noble gases on the periodic table are formed of completed P-orbital sets. These inert elements define the qualities of each atom and its ability to form bonds. Once the noble configuration forms, all subsequent atoms retain the same inner orbital structure, with only the electrons at the highest energy level available for bonding.
This 4D geometric perspective provides a more comprehensive explanation for the structure of the periodic table than traditional quantum mechanics, which is often only applicable to hydrogen-like atoms. The conclusion is that the 4D sphere and its derived counterpart, the torus, are the geometric forms that concentrate vacuum energy through the nature of their 4D rotational spin.
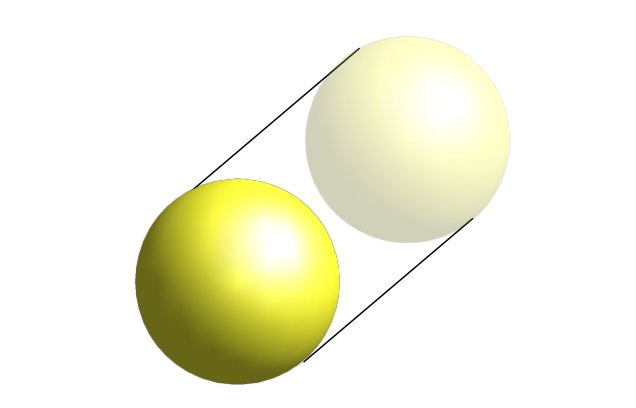

This also explains why the electron radius has never been established — the electron is not a particle but a 4D field. It also provides a reason why the number of protons and electrons in an atom is always equal: rather than being separate entities, the two are unified by the 4D field. Protons also exhibit half-spin but are composed of quarks with 1/3 and 2/3 spin values, providing a mechanism for greater concentration of vacuum energy within the nucleus. The 4D Aether and the article on 4D matter explore how this gives rise to mass and charge in the universe.
Orbital configuration
The first 20 elements are formed with just S and P orbital types. The first shell forms from a single S-orbital. The second shell comprises another S-orbital followed by three P-orbitals, completing at Neon (10). The third shell repeats this pattern at Argon (18). In the 4th shell, an S-orbital electron pair produces a torus field around the whole structure. This configuration maps onto the Metatron's Cube mandala, an extension of the Flower of Life.
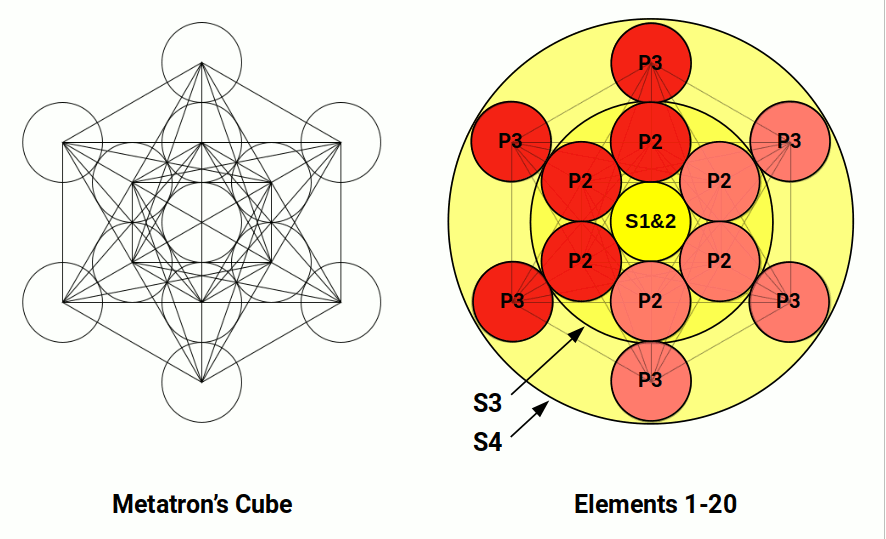
At this point the pattern of orbital formation changes. Instead of forming another P-orbital set in the 4th shell, a set of D-orbitals forms in the 3rd shell. Unlike P-orbitals, D-orbitals require an S-orbital in the shell above before they can appear — a fact obscured by the periodic table placing the first D-orbital shell in the 4th row rather than the 3rd. A similar constraint applies to F-orbitals, which only appear in the 4th shell once S and P-orbitals form in the 5th shell above.
Current atomic theory offers no explanation for this ordering, but geometry does. S and P-orbitals are derived from the 4D sphere; D and F-orbitals are mapped onto the 2D plane. In D-orbital sets, two out of four lobes are filled simultaneously; in F-orbital types, three out of six. The S-orbital hypersphere in the shell above acts as an energy container. The three P-orbitals align on the x, y, and z axes, forming an octahedron — the Platonic solid that is the dual of the cube. The P-orbitals occupy the centre of each face of the cube, leaving the corner spaces that D-orbital electrons can then fill.
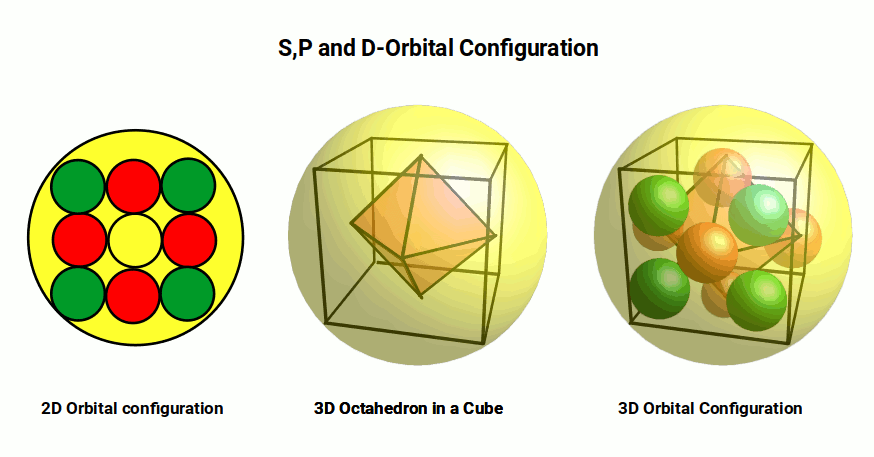
A similar process governs F-orbitals, which form a hexagon in 2D and a cuboctahedron in 3D. The cuboctahedron — sometimes called the vector equilibrium because each corner is exactly equidistant from the centre and from adjacent corners — is the most efficient shape for nesting spheres in 3D space. When the cube combines with the octahedron, the midpoints of each side connect to form the cuboctahedron. This provides a simple geometric reason why F-orbitals can only appear once the octahedral P-orbitals are established in the shell above.
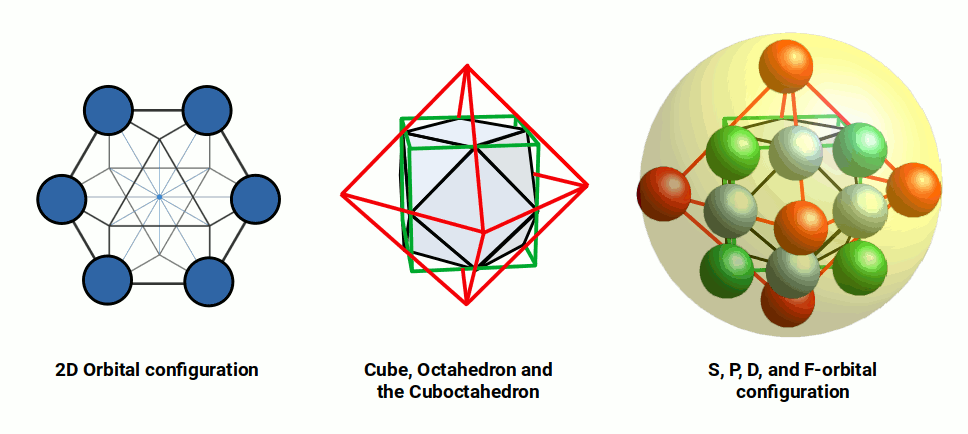
In summary: S and P-orbitals appear sequentially, with P-orbitals outside the S-orbital sphere. D-orbitals appear after the P-orbitals but must be enclosed inside an S-orbital in the shell above. F-orbitals appear after the D-orbitals and are enclosed in a set of S and P-orbitals in the shell above. This can be represented as a simple 2D map.
Conclusion
This model resolves numerous quantum paradoxes. It explains the quantised nature of electron spin, why there are only four types of orbital, and why those types appear in the order they do. The configuration of orbitals is explained by the evolution of geometric forms following the principles of 2D and 3D geometry, without the need for complex mathematics.
The 4D geometric perspective offers a new approach to the atom that can explain the most intricate details of the electron cloud in alignment with traditional quantum probability calculations — and goes further by providing a logical, geometric reason for each structural feature. For a deeper exploration of the underlying theory, see 4D Aether.
FAQ
Why are there only four types of electron orbital?
The four orbital types — S, P, D, and F — correspond to the only geometric primitives that arise from the two regular 2D tessellations using two colours: the square plane and the triangular plane. After the F-orbital (hexagon), no further regular tessellation is possible with two colours, which is why no higher orbital types have ever been observed.
How does 4D geometry explain quantised electron spin?
Electron spin can be modelled as two 2D planes that swap places in 4D space, similar to a 4D hypercube rotating on its W-axis. The two orientations correspond to up and down spin. Because the swap is discrete — the plane either occupies 3D space or it does not — the spin is inherently quantised, with no intermediate states.
Does the 4D sphere and torus model apply to D and F-orbitals as well?
Yes. The D-orbital set corresponds to a 4D hypercube (tesseract), represented as two nested cubes. The F-orbitals correspond to the 24-cell octaplex, whose geometry is expressed by the Cuboctahedron combined with its dual, the rhombic dodecahedron.
Why do D-orbitals only appear after an S-orbital forms in the shell above?
S and P-orbitals are derived from the 4D sphere; D and F-orbitals are mapped onto the 2D plane. The S-orbital hypersphere in the shell above acts as an energy container. The three P-orbitals align on the x, y, and z axes forming an octahedron, leaving corner spaces that the D-orbital electrons can then occupy. Without that containing shell, the 2D-plane orbitals cannot form.


