The electron cloud is formed of four types of orbitals S, P, D, and F. The most simple of these is the S-orbital type which appears as a simple sphere. In this article — part of the Atomic Geometry series — we explore the various S-orbitals that appear in the stable elements on the periodic table, and examine their radii from the perspective of geometric ratio.
Key Takeaways
- The S-orbital is the simplest electron orbital type — a spherical shell that appears as the first orbital in every atomic shell.
- Standard quantum models (Bohr radius, Van der Waals radius) fail to predict experimentally measured atomic radii — the Bohr model is over 100% out for hydrogen alone.
- A geometric model based on √2, √3, and √1.5 (the helium radius) predicts all 12 stable S-orbital element radii within the experimental margin of error.
- The Jitterbug transformation and the cube-to-circumsphere ratio (√3÷2) connect successive S-orbital radii through simple geometric relationships.
- The helium atom uniquely satisfies a three-dimensional "squaring of the circle" — its spherical surface area equals that of a cube with face area π.
What is an S-orbital?
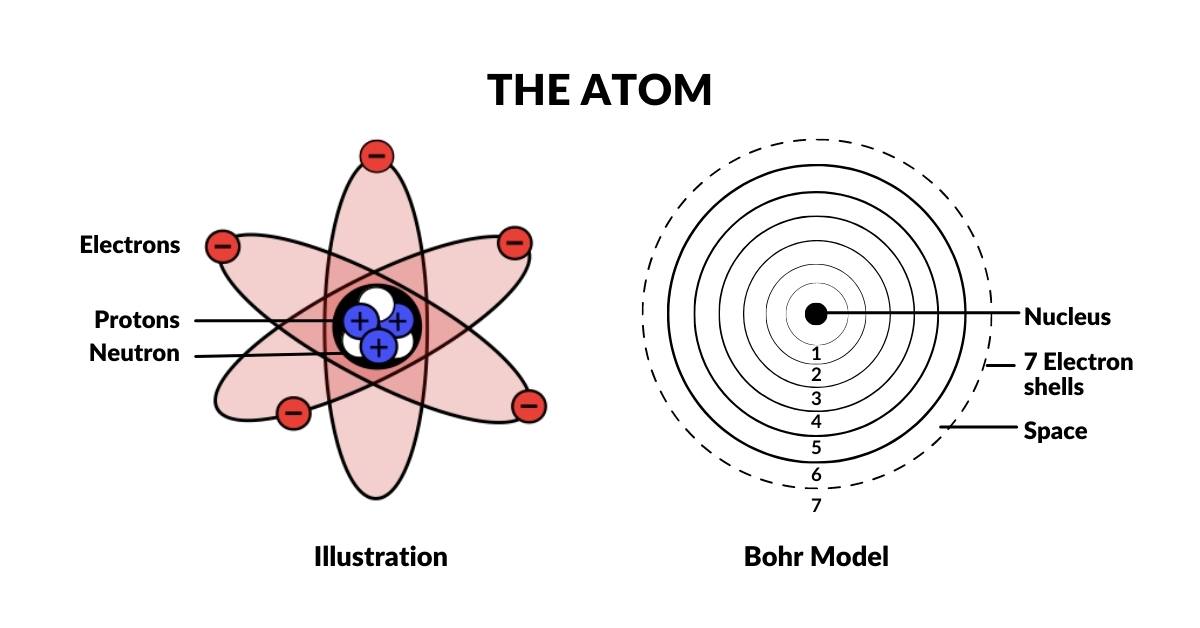
All atoms are comprised of a nucleus formed of protons and neutrons, surrounded by an electron cloud made of electrons. When the structure of the atom was first discovered, it soon became apparent that electrons can only appear at distinct distances from the nucleus. Furthermore, these electron shells come in four types, S, P, D, and F. Each of these exhibits a specific type of geometric configuration. The S-orbital type is the only one that appears spherical in nature.
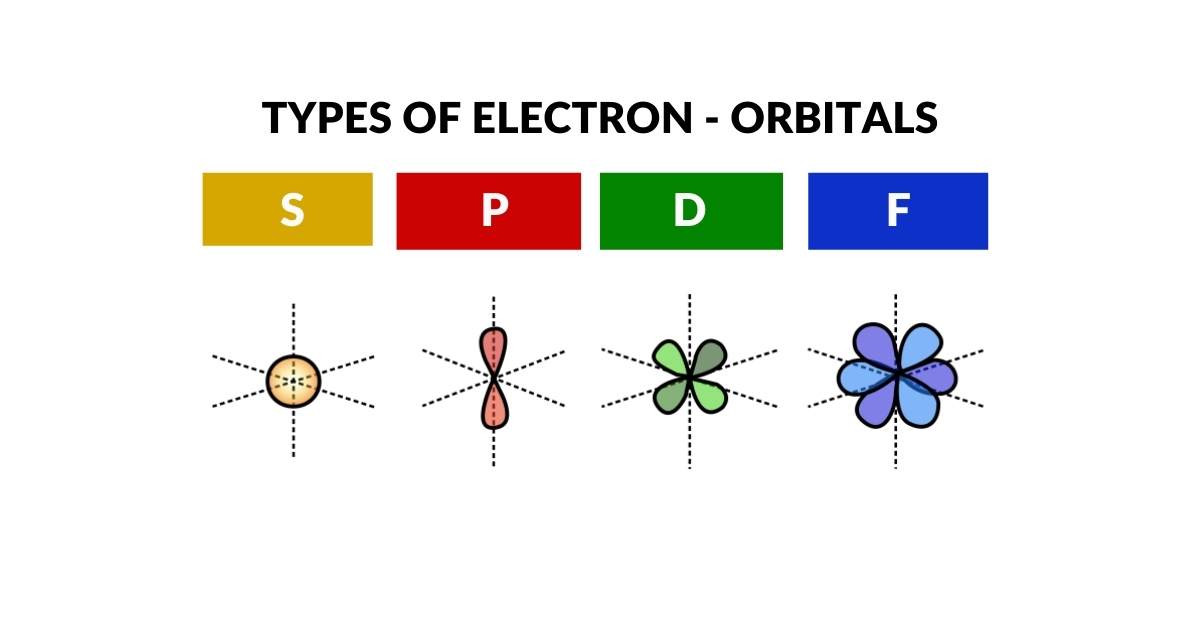
Each orbital is composed of two electrons, which exhibits either an up or down quantum spin value. The S-orbitals are always the first to appear in each shell, and lie at the heart of many atomic bonds. Except for Helium, all S-orbitals are found on the far left of the periodic table. These appear in group 1 and 2. Group 1 elements are formed of a single electron in the S-orbital shell, whilst group 2 have two. Helium appears on the far right in group 18, as it is a noble gas, which means it does not make bonds with other atoms.
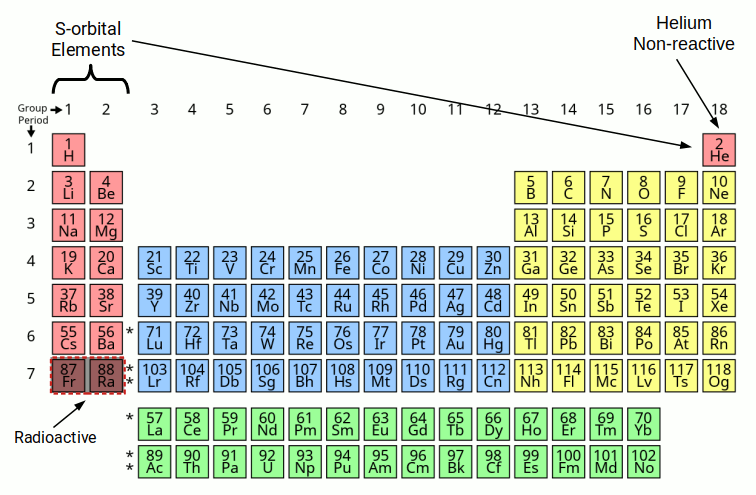
Except for P-orbitals, (yellow), all D (blue) and F (green) orbital elements exhibit an S-orbital in the shell above. These are able to form bonds with other atoms as they are furthest away from the nucleus. This makes S-orbital electrons key to the formation of many molecules and compounds.
What are electron Shells?
Most people are aware that the electron that surrounds the atom can only appear at specific distances from the atomic nucleus. These are called shells. Each shell of the atom can only contain a certain number of electrons before it becomes full. After this, the electrons begin to fill the next shell. The first two electrons to fill a shell are always the S-orbital type. There are only 6 S-orbitals that arise in the complete set of stable elements. The 7th shell is radioactive. This is sometimes referred to as the Bohr Model of the atom.
The radius of an atom is often defined in Angstroms (Å). One Angstrom is equivalent to 100 picometres. You might think that the S-orbitals should get further away from the nucleus as the shell number increases. However, the radius of the atom grows larger and smaller in a seemingly unpredictable way. The first atom, Hydrogen (1), has a radius of only 0.25 Å. When the first S-orbtial completes with Helium, the radius increases to around 1.2 Å. The 3rd element, Lithium (3), expands to an even greater size with a radius of 1.4Å. But then the next element, Beryllium (4), completes the second S-orbital, where the radius is reduced to just 1.05 Å. Aside from the first 2 elements, the other S-orbitals generally follow the same pattern, whereby the first S-orbital comprised of a single electron is larger than the next one formed of two electrons.
Horizontal Axis = Shell Number, Vertical Axis = Radius in 100 picometre scale
The first shell
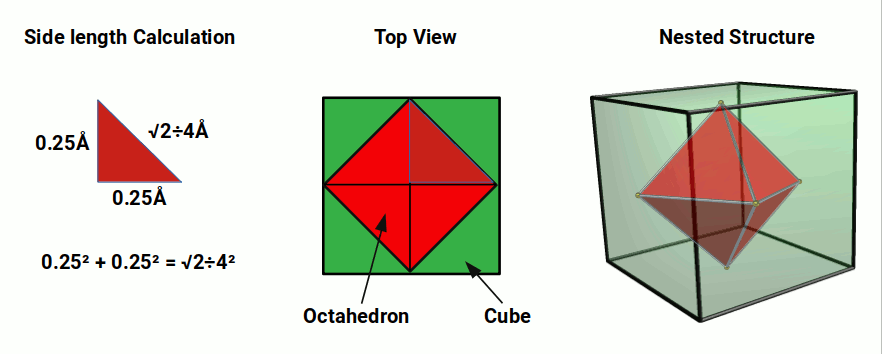
The first shell of the atom is the only one to be comprised of a single S-orbital shell. This begins with Hydrogen (1), comprised of a single proton and electron. It often forms a bond with another hydrogen atom to from the H₂ Molecule. This is found to comprise around 75% of matter in the universe. Hydrogen has an experimentally measured radius of around 0.25Å. This means the S-orbital will fit inside a cube of space with a side length of 0.5 Å.
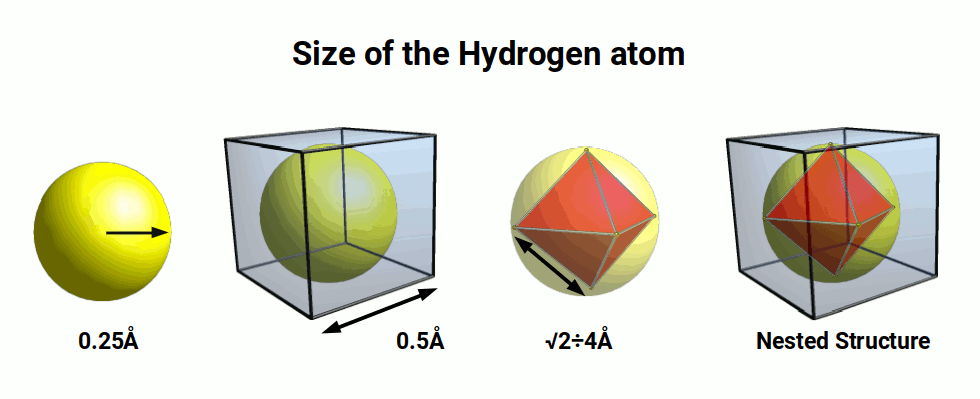
The Octahedron (red) in the image above is nested inside the cube. The hydrogen sphere sits in the space between the octahedron and cube. The side length of the octahedron is calculated using the Pythagorean theorem of right-angled triangles. The sphere’s radius forms the base and height of the triangle, to define the diagonal hypotenuse, which is the side length of the octahedron. Therefore, in order to ascertain the side length of an octahedron from the radius of a sphere that encompasses it, we can multiply the radius by √2.
The next atom to appear is Helium. Its experimentally measured radius is considerably bigger than hydrogen, with a value of around 1.2Å. This can partly be attributed to the fact that it has a nucleus that consists of two protons and two neutrons. This is about four times the size of the hydrogen atom.
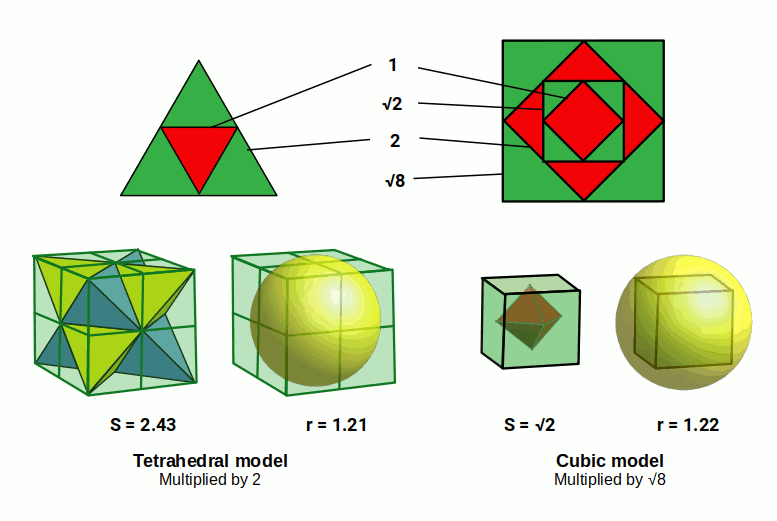
These four nucleons, can be arranged into a tetrahedral formation. If each projects a sphere the same size as the hydrogen (1) atom, the side length of the tetrahedron will be 1.72Å. When the tetrahedron is nested inside a cube, each side is falls across the diagonal of each of the cubes faces. The ratio of this diagonal to the side of the cube is 1:√2. By dividing 1.72Å by √2, we arrive at the side length of 1.21Å for the cube.
This very close to the radius of the helium atom. However, the radius of a sphere is only half the distance of the cubes side. Therefore, we need to double the size of the cube in order to create the correct size that will nest the helium sphere. This means that each of the original spheres that formed the tetrahedron is doubled in size. Each will have a radius of 0.5Å, and so a Diameter of 1Å.
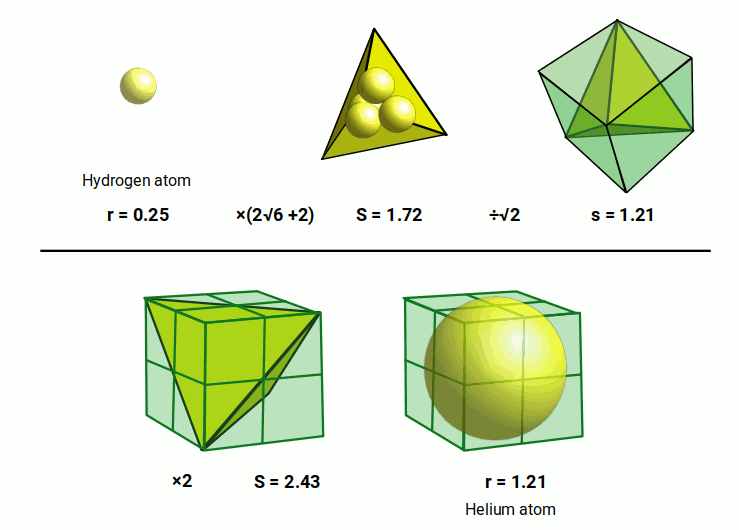
We should expect that a nucleus with four times the number of nucleons would create a larger atomic radius. However, in this tetrahedral model, each sphere doubles in size. The Helium radius is around 2√6 (4.9) times bigger than Hydrogen. This expansion rate of 2√6 is geometrically associated to a cube. When the hydrogen radius of 0.25Å is multiplied by √8, the result is a radius of √0.5. This larger sphere can be placed inside a cube with a side length of √2. Around this cube, a second sphere can be positioned that will have a radius of 1.224Å or √6÷2.
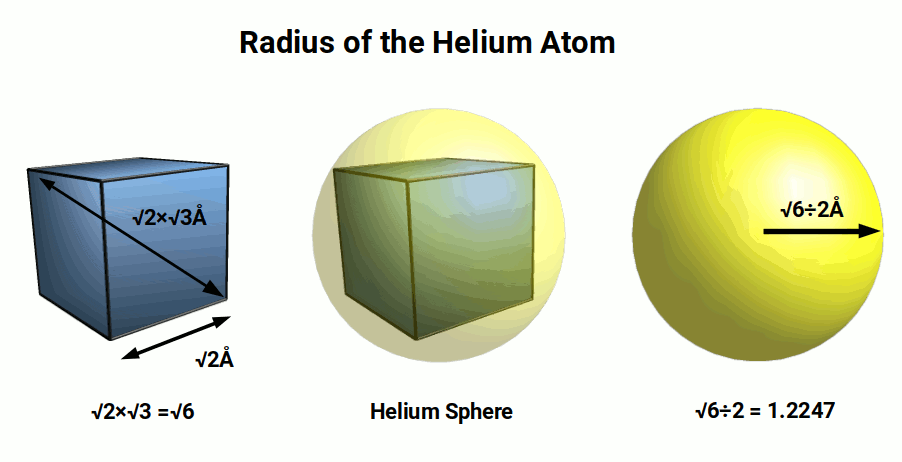
The cube has corner points that increase the size of the sphere by √3. This is because a cube with a side length of 1 will exhibit a diagonal that is √3 from each of its furtherest corners. Therefore, to find the radius of the sphere that encompasses the cube, its side length can be multiplied by √3÷2.
This cubic transformation of the hydrogen radius into helium can be mapped by a geometric process called the Jitterbug. First conceived by Buckminster Fuller, the Jitterbug is a geometric transformation that changes one polyhedron into another through rotation. Again the side length of the octahedron that sits at the centre of the helium atom is multiplied by √8 to create an Octahedron with a side length of 1Å. Each face rotates, expanding to form an icosahedron, followed by a cuboctahedron. The cuboctahedron combines the faces of the octahedron and cube into a single form. By adding 8 pyramids to its triangular faces, the cube is formed, with a side length of √2. The sphere that encompasses the cube will have the same radius as the helium atom.
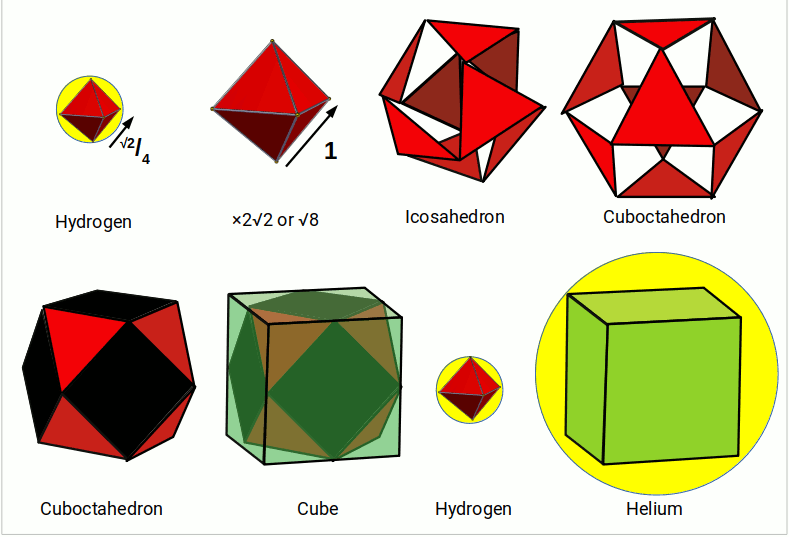
Just as the four nucleons of Helium can be arranged into a tetrahedron, they can also be aligned into a square. Again, let us consider that each creates a field with a radius of 0.25Å. Therefore, the total side length of the square will be 1Å. This square can be placed inside a cube, nested inside a cuboctahedron with the same side length. Each of the cubes corners will touch the centre of the Cuboctahedrons triangular faces. When the corners are added to the cuboctahedron, is creates a cube with a side length of √2. This defines the radius of the Helium atom.
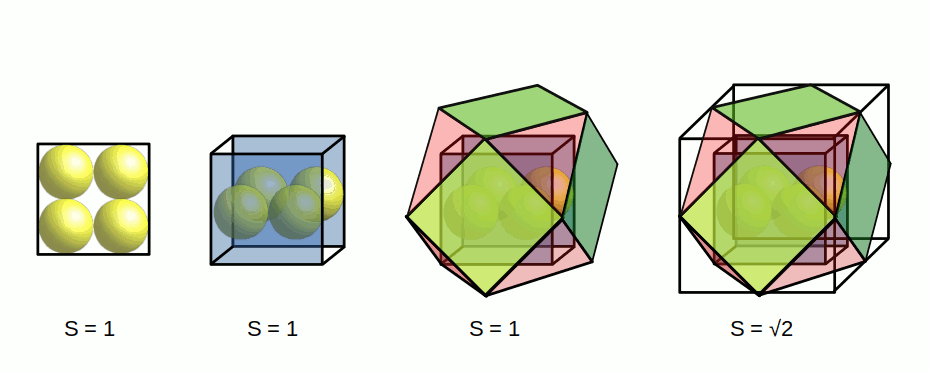
These examples show two types of geometric transformation, which demonstrate the relationship between the radius of hydrogen (1) and helium (2). The expansion through the tetrahedron demands a doubling of the fundamental radius to create a tetrahedron of the correct size. The cubic transformation creates a square with a side length of 1, which expands through the jitterbug to form a cube of side length √2. Finally, this is multiplied by √3÷2, to find the radius of the sphere that encompasses the cube. The doubling expansion of the tetrahedron is expressed by nested two triangles. The cubic transformation is found in a series of squares nested at 45°, which expands at the rate of 1:√2.
The Helium radius is contained between the inside of the cube generated by the tetrahedron, and outside the cube generated by the octahedron. This geometric interpretation of the atomic radii is much more accurate than the calculated values based on traditional quantum theory. Present predictions are based on two different metrics. The Bohr Radius is derived from the Bohr model of the atom, whereas the Van der Waals tries to treat the atom as a solid sphere.
Both place the hydrogen atom as being much larger than experimental measurements. Helium begins to approach the predicted size according to Van der Waal radius, however the Bohr radius predicts the atomic radius should get smaller. When we compare the experimentally determined radius with the geometric model, we find the match is well within the 5 picometre margin of error.
The Tetrahedral and Cubic Model follow the predicted experimental radii so closely, it is difficult to discern the difference.
The traditional concept of the atom is based on the nature of charge. The positively charges proton attracts the negatively charged electron. When the electron reaches its ground state (first shell), then it reaches an electrical equilibrium with the proton. The neutron does not exhibit a charge, and so is not supposed to attract or repel the proton or electron. This model gives the proton and electron the same amount of charge, defined by the elementary charge constant, e.
Yet the erratic growth of the helium radius can not be explained using this model. In fact, quantum mechanics is only valid for hydrogen like atoms. Yet, even this statement is not particularly accurate. The Bohr radius is over 100% out with a suggested radius of 0.53Å, compared to the experimentally determined radius of 0.25Å. The Van der Wall radius provides an even poor prediction of 1.2Å, which is actually the radius of the Helium atom. According to the Bohr model, the radius of helium should diminish as the nucleus grows in size. This should give Helium a smaller radius of 0.31Å. Clearly this is not the case. The Van der Wall radius predicts a size of 1.4Å, which is actually the radius for the third element, Beryllium.
The truth is that the current mainstream models of the atom cannot predict the experimentally verified of the most simple atomic radii. The idea that the universe can be broken down into specific units of fixed electrical charge should see an atom whose radius increases in incremental steps. However, this is not the case. The rapid radial expansion of Hydrogen to Helium can only be explained using a geometric model of the electron cloud.
The first Noble Gas
Hydrogen and Helium complete the first shell of the atom. After this, subsequent electrons will begin to fill the next shell. Helium (2) is completely inert. These elements are call Noble Gases, which do not form bonds with other atoms. They are key to the structure of the electron cloud. Once a noble gas is formed, only electrons from a higher energy level can be used to make bonds. These subsequent electrons are found in the outermost valance shell of the atom. All noble gases are found at the far right of the periodic table in group 18. Helium is the only inert gas to be formed from S-orbital electrons. All other Nobel Gases are created by a complete set of three P-orbitals.
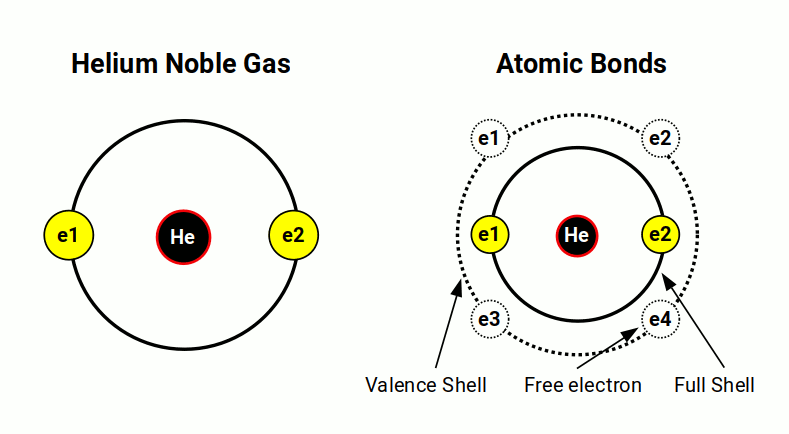
Present atomic theory does not offer any kind of logical explanation as to why helium should be a noble gas. It only points to the fact that shells filled with the maximum number of electrons will be inert. Can the geometric solution to the electron cloud does offer a more explicit explanation?
Helium has a radius of around √6÷2. Its volume can be calculated from the square of its radius with the equation r³ × 4/3π. This provides a result of √6π. We can also calculate its surface area by the equation r² ×4π. For a sphere with a radius, √6÷2 will have a surface area of 6π. The helium atom has a diameter of √6. This means that when diameter is multiplied by its volume, the result is the surface area of the sphere. This quality is unique to this particular sphere. As the volume is calculated by r³, the surface by r², from a radius of r1, we find a perfect union of these numerical powers.
The surface area of this hydrogen sphere is 6π. This means it has the same surface area as a cube, where each face has an area of π. This gives the cube a side length of √π.
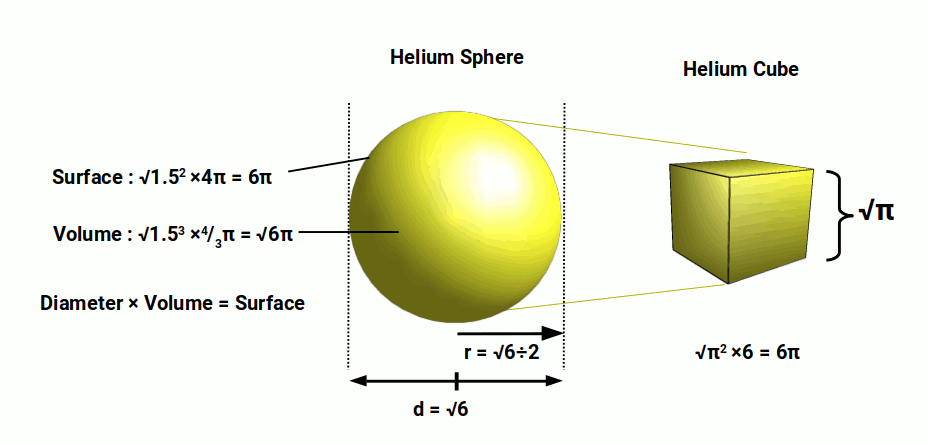
The area of a circle is determined by the equation πr². A circle with a radius of 1 will therefore have a surface area of π. This is the same value as the surface of a square with a side length of √π. In geometry, this is known as the squaring of the circle, which has remained a curious enigma for both geometricians and mathematicians. Yet, the radius of the Helium atom provides us with a three-dimensional solution.
The unification of the surface of a sphere, to that of the cube, expresses two kinds of space. Cubes can fill a 3D space, whereas spheres leave small gaps. If a sphere is compressed into a cube, it will maintain its surface area. However, now it will be able to fill a 3D space.
The nature of cubing the sphere, and the number √1.5 is also found in our wave solution to the photoelectric effect. This value is found as (√c²×G)÷2, where c is the speed of light and G is the gravitational constant. When divided by the work function of a particular material in EVs, the result is the threshold wavelength that initiates the photoelectric effect. Whilst the work function of different atoms varies for each atom, its relationship to the threshold frequency remains consistent. What we find is that Helium has the same relative distance as its radius. Could this for the first time offer a reason as to why it is a noble gas?
The first 4 S-orbitals
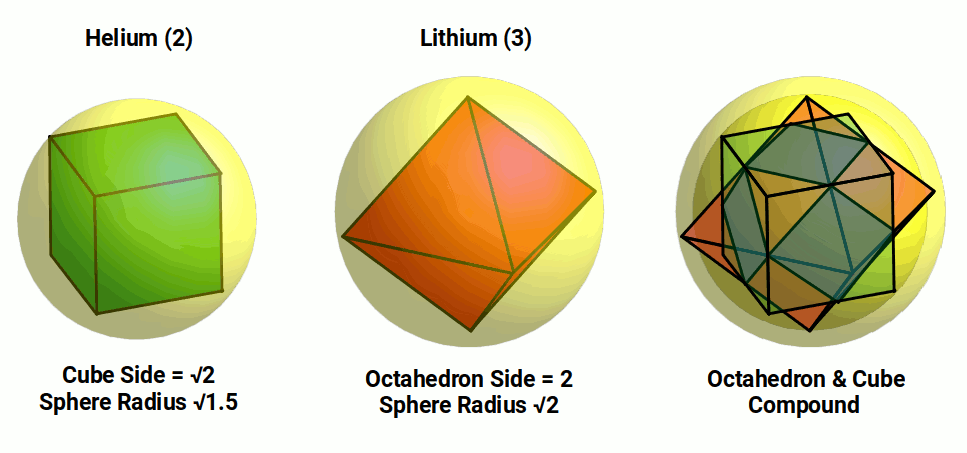
Once the first shell completes, electrons begin to fill the second shell. The third and fourth elements, Lithium (3) and Beryllium (4), which comprise a 2nd S-orbital. Lithium (3) is larger than helium, with a radius of around 1.4Å, whereas Beryllium is smaller, with a radius of only 1.05Å.
The radius of Lithium is extremely close to √2 (1.414). This means that is can be placed around an octahedron with a side length of 2. When we combine this octahedron with the cube, side length √2, which defines the Helium atom, it generates a compound of the cube and octahedron. The sides of each polyhedron meet at the midpoint, which defines the cuboctahedron.
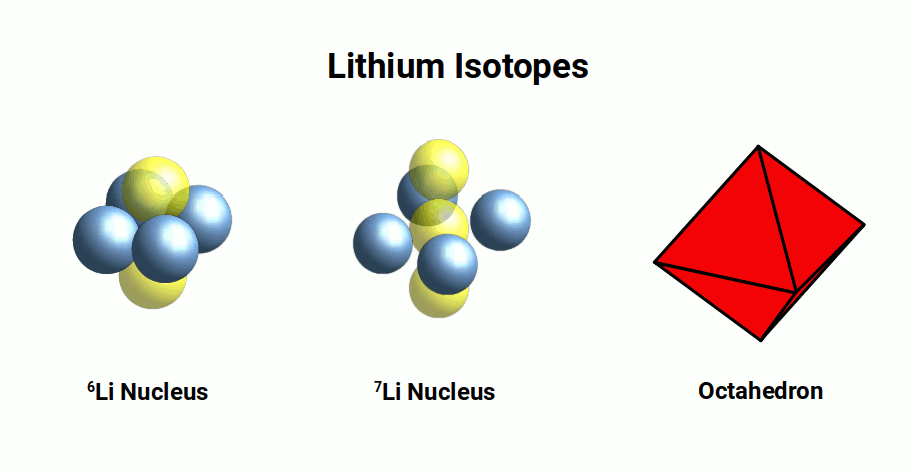
This radius of the Lithium atom forms the basis for the majority of D-orbitals elements, which tend to exhibit a radius between 1.35Å and 1.45Å. Lithium appears as two different stable elements. One with 3 Neutrons and another with 4 Neutrons. These are called isotopes. The most predominant is 7Li, with 4 neutrons and 3 protons. When arranged in 3D space, the nucleus forms an octahedron.
The corner points of the compound of the Cube and Octahedron can be connected to create the 12 faces of the Rhombic-Dodecahedron. This polyhedron is the template for the 4D hypercube. Just like the cube, the rhombic-dodecahedron can fill space perfectly.

The 4D Hypercube is sometimes represented as one cube nested inside another. Unlike regular 3D objects, 4D polytopes each exhibit a 4th dimensional w-axis, which is akin to the nature of time. When rotated on this time axis, each cube swaps places. The same structure is also found in our solution to the 4D Aether.
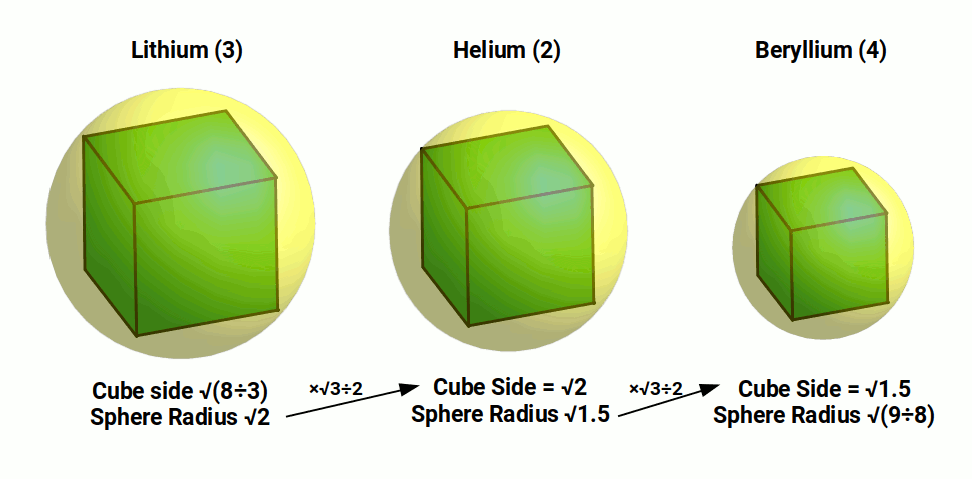
Thus far, the radius of each Atom has increased in size as the atomic number increases. This trend changes with the next atom, Beryllium (4), which has a much smaller radius of 1.05Å. If a cube has the side length of the radius of the Helium atom, √1.5Å, then the sphere that encompasses the cube will have a radius of 3√2÷4, or 1.06Å, very close to the radius of Beryllium (4).
This produces an interesting geometric relationship between the radius of Helium (2), Lithium (3), and Beryllium (4). Starting with the largest element, Lithium, with a radius of √2, we can represent this as a sphere that encompassed a cube. This cube will have a side length of √(8÷3). When we multiply √2 by √3÷2 to obtain the side length of the cube, the result is √6÷2, which is the radius of the Helium atom. We can repeat the process and multiply √6÷2 by √3÷2 and get the radius of the Beryllium atom, √9÷8, or 1.06Å. Therefore, these atoms are all in perfect geometric ratio to each other, based on the side length of the cube to the radius of the sphere that encompasses it.
This geometric interpretation gives a radius for Beryllium (4) of 1.06Å is exactly 3√2 times larger than the hydrogen atom (1), with a radius of 0.25. When 0.25 is multiplied by 4√2, the result is the radius of Lithium (3). Therefore, the radius of both Lithium and Beryllium are in ratio to the Hydrogen radius through the √2.
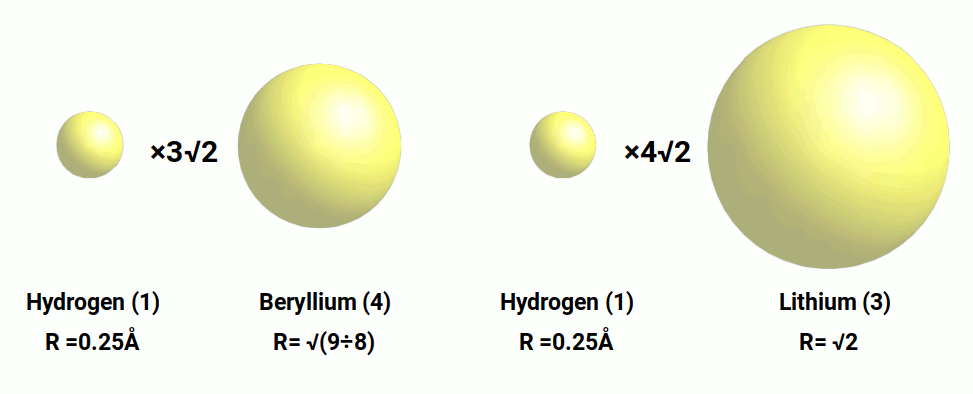
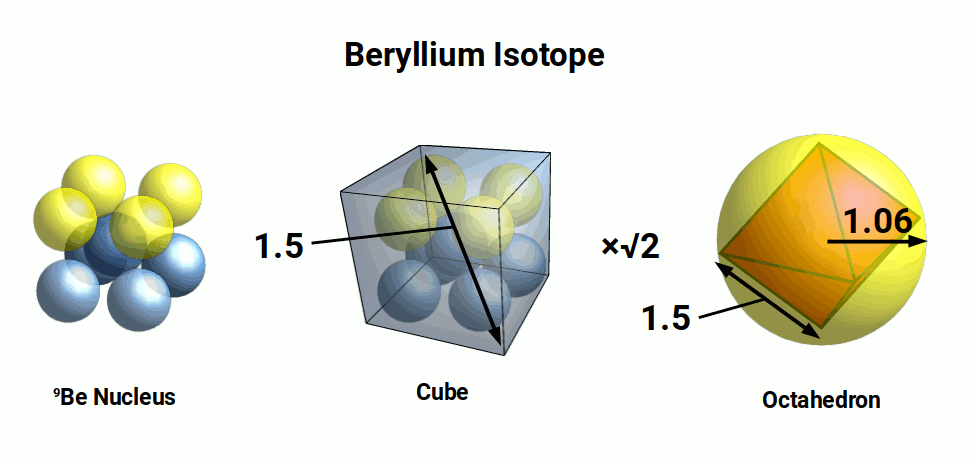
Beryllium (4) only has one stable isotope comprised of 4 protons and 5 neutrons, (9Be). These can be arranged into a cube, with the additional Neutron placed at the cubes centre. Given that each nucleon produces a sphere of radius of 0.25Å, the distance across the diagonal of the cube will measure 1.5Å, giving a radius of 0.75Å. When multiplied by √2, the result is the diameter of the Beryllium atom (1.06Å). Additionally, we find that this radius is produced by a sphere that encompasses an octahedron with a side length of 1.5Å.
The first 4 elements are all found to exhibit radii that are in ratio to each other. Again, we can compare the geometric model to the experimental and theoretically determined values.
We can see that the geometric view provides a very close fit to the experimental radii. The Bohr Radii expands dramatically from helium to lithium, overshooting the experimental values by 0.27Å. When it comes to Beryllium (4), the Bohr model does come closer, with a radius of 1.12Å, however that is still 0.06Å larger, which is just outside the experimental error of ±0.05Å. The Van der Waals radius is much larger, overshooting the experimental values by 0.41Å for Lithium (3) and 0.48Å for Beryllium (4). Only the geometric model is able to predict the radii within the expected experimental error.
S-orbitals in the 3rd shell
After Beryllium (4), the second shell completes with a set of 6 P-orbital elements, up to elements 10, Neon. After this, the 3rd shell begins to fills with two S-orbital Electrons, Sodium (11) and Magnesium (12). Both have radii larger than the previous S-orbitals, with the largest being Sodium (11) 1.8Å compared to 1.5Å for Magnesium (12). The radius of these atoms are in ratio to the Beryllium (4) radius of 1.06Å, the last S-orbital element of the previous shell, through the geometry of the cube.
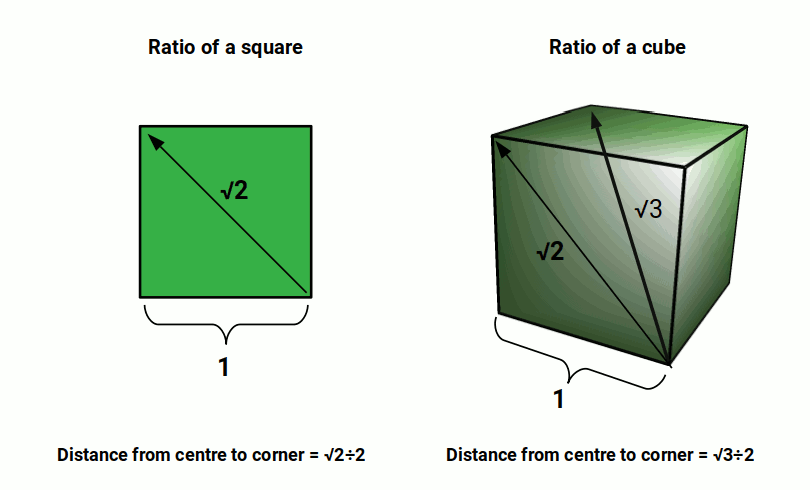
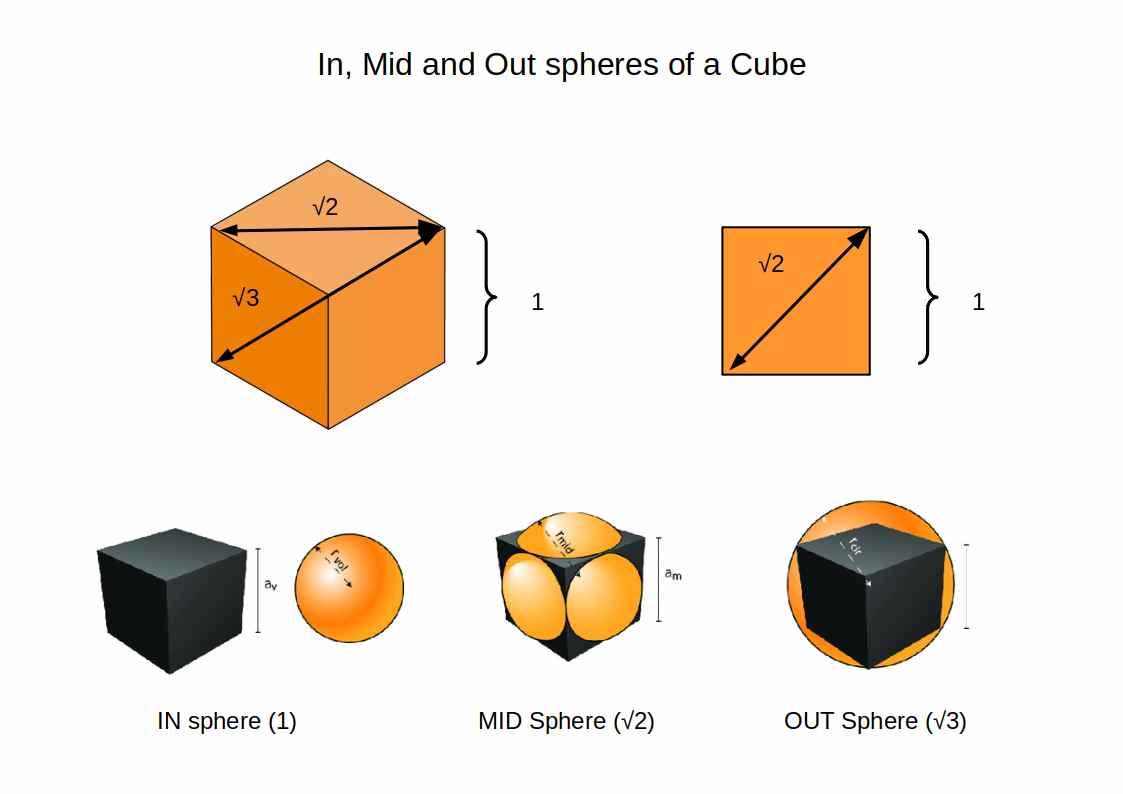
A cube exhibits three kinds of sphere, Insphere, Midsphere, and Outsphere, sometimes referred to as the circumsphere. The Insphere appears inside the cube, touching the centre of each of its faces. The Midsphere is larger by a factor of √2, and touches the midpoint of each side of the cube. The Outsphere is larger by a factor of √3, and encompasses the cube, touching each of its corners.
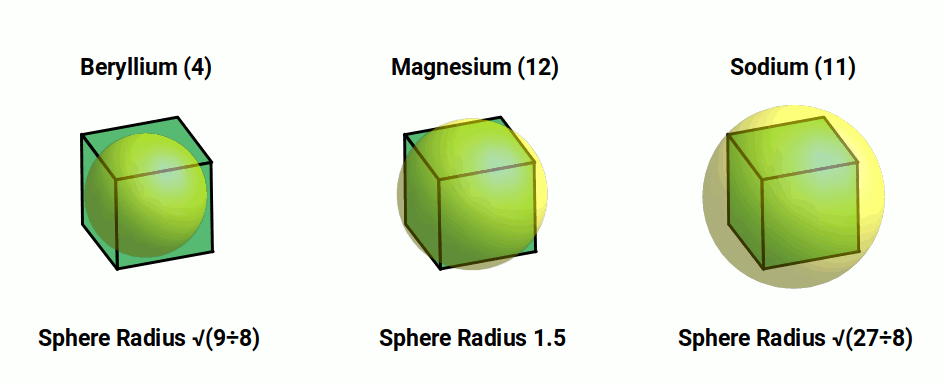
Beryllium (4) sphere has a radius of √(9/8). We can place this inside a cube, that will have a side length of √(9/2), double this value. If we multiply the radius of Beryllium by √3, the result is 1.83Å, close to the experimental radius of Sodium (11). Whereas multiplying the radius by √2 produces 1.5Å, the radius of Magnesium. Therefore, all the radii of these three elements falls into the In, Mid, and Out sphere of the cube.
Again, we find that this relationship can be expressed through the jitterbug transformation. Previously, we saw that the Beryllium sphere will encompass an Octahedron with a side length of 1.5Å. This expands through the jitterbug to produce a cuboctahedron with the same side length. The magnesium sphere will fit perfectly around this form. When the corners are added to the cuboctahedron to form a cube, so we find the radius of the Sodium sphere.
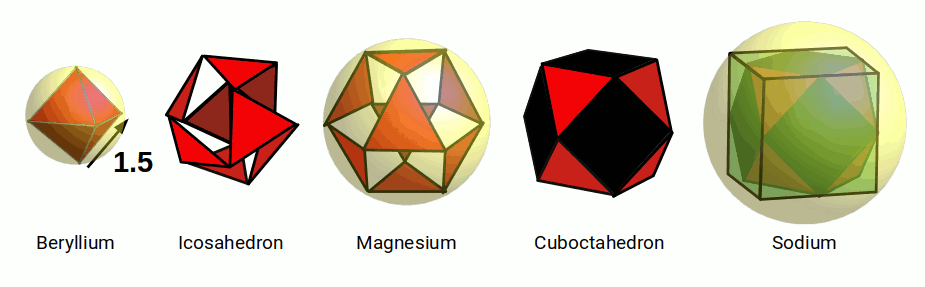
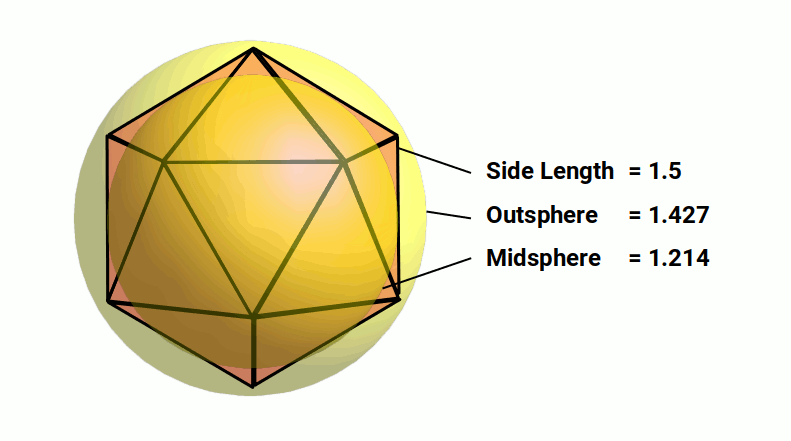
The Icosahedron appears in the transformation of the octahedron into the cuboctahedron in the jitterbug transformation. Like the cube, also exhibits an In, Mid, and Out sphere. In the previous model, the octahedron exhibits a side length of 1.5Å, which can expand into the icosahedron through the jitterbug. With a side length of 1.5 the radius of its Midsphere will be 1.214 very close to the radius of Hydrogen. The Outsphere will have a radius of 1.427 which is close to the radius of Lithium.
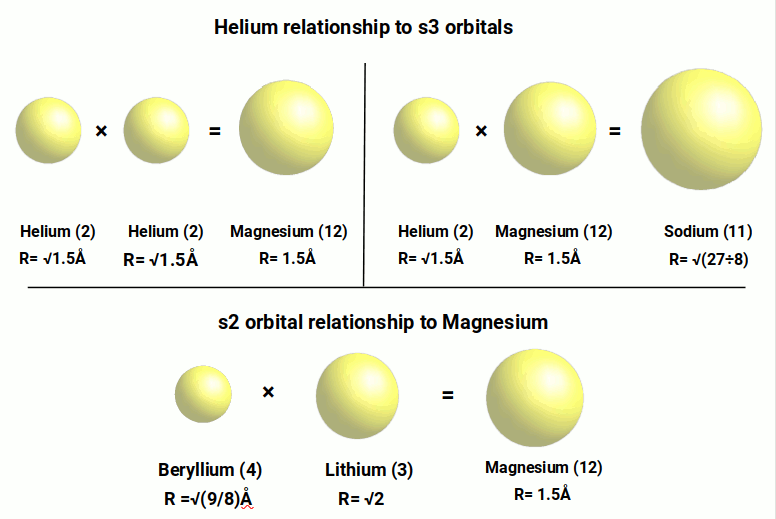
We also find that the Helium radius of √1.5Å is in ratio to the third pair of S-orbital elements. When squared, the result of 1.5Å is the radius of Magnesium (12). When both of these are multiplied together, √1.5Å × 1.5Å, the result is the radius of Sodium (11). The radius of magnesium can also be calculated by multiplying the radius of the S-orbital elements in the 2nd shell. Lithium (3), with a radius of √2Å, is multiplied by Beryllium (4), radius √9÷8Å, to produce a radius of 1.5Å.
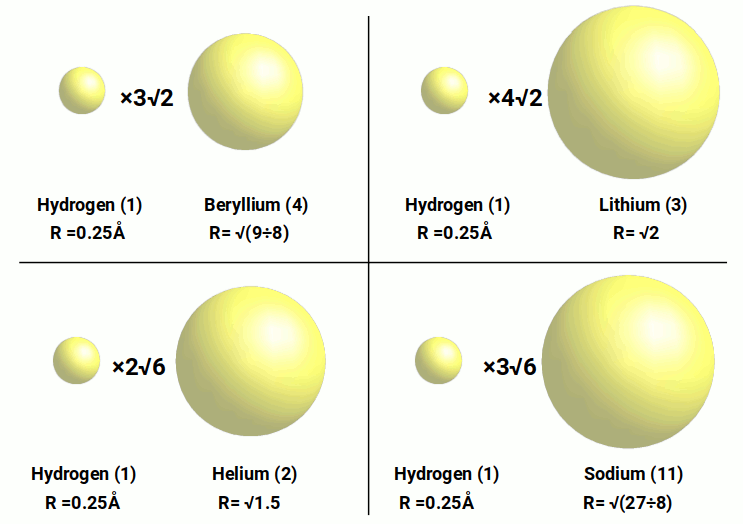
Finally, the hydrogen radius of 0.25Å is multiplied by 6 to produce the radius of Magnesium, and by 3√6 to produce the radius of Sodium. This reveals another simple pattern. When the Hydrogen radius is multiplied by 2√6, the result is the radius of Helium, and when multiplied by 3√6, the result is the radius of Sodium. Previously we saw that the Hydrogen radius multiplied by 3√2 and 4√2 will define the radius of Beryllium (4) and Lithium (3) respectively. A similar relationship defines the radius of Helium and Sodium. It is worth noting that √6 is found my multiplying √2 by √3 and is the diagonal of a cube with a side length of √2, found in the compound of the Cube and octahedron, which produces the Rhombic-Dodecahedron in the previous section.
The geometric model shows a relationship to the various atomic radii, based on the √2 and √6. We can compare the result to the experimental and calculated radii. The Bohr model comes close to the expected values, with a radius of 1.9Å for sodium and for Magnesium, it falls 0.5Å below the experimental value, which is just on the cusp of the experimental margin. The Van der Waals radius overshoots by 0.47Å for sodium and 0.23Å for magnesium. However, this is expected as the model is based on the extreme limitation of the electron cloud. Yet the geometric model is only +0.3Å over for Sodium and an exact match for Magnesium, which is within the margin of experimental error.
This is about as close to an accurate prediction for the S-orbital elements as the Bohr radius gets. After this the level of inaccuracy increases after we shall see when we examine the remaining S-orbital elements.
S-orbitals in the 4th shell
After Magnesium, another set of six P-orbital elements form to complete the next noble gas, Argon (18). Then, the next set of S-orbitals form in the 4th Shell. Potassium (19) and Calcium (20) have a radius of 2.2Å and 1.8Å respectively. Notice that sodium (11) and Calcium (20) have the same radius. Yet, the number of protons and neutrons in these two nuclei is vastly different. The most stable isotope of Calcium has 20 protons and 20 neutrons, whereas Sodium has 12 Neutron and 11 protons. We should think that the Calcium radii should differ if the electromagnetic model of the atom were true, as it contains an additional 17 nucleons.
Again, we find there is a geometric relationship between the two radii of the S-orbital atoms in the 3rd and 4th shell. As noted previously, when the Magnesium (12) radius is multiplied by √1.5 the result is the radius of Sodium (11). Therefore, the same ratio also applies to the Calcium (20) Atom. If we multiply this radius again by √1.5, the result is 2.25Å, only +0.3Å above the experimentally determined radii. As √1.5 is the radius of Helium, the radius of the s3 and s4 elements can be found by raising √1.5 to the power of 2, 3 and 4.

Again, we can compare these geometric radii to the experimental and calculated radii. What we find is that the calculated values predict a larger radius than is expected. The fact that Sodium and Calcium exhibit the same radius cannot be accounted for by the calculated values. The Bohr Radius is +0.23Å and +0.14Å greater than the experimentally determined radii and outside the margin of experimental error. The Van der Waals radius expresses an even larger value of +0.55Å and +0.51Å greater. Only the geometric model produces values within the expected margin of experimental error.
Once the S-orbitals have completed in the 4th shell, the first set of 10 D-orbital elements begins to form in the 3rd shell below, up to Zinc (30). These elements generally exhibit a radius of around 1.4Å or 1.35Å. The fact that many of these elements exhibit the same radius cannot be explained by the standard model of quantum mechanics. However, from the perspective of the geometric model, we notice that 1.4Å is the same radius as Lithium (3). The second radius of 1.35Å can be defined by dividing the Lithium radius by the Beryllium radius, which produces the result of 1.33Å.
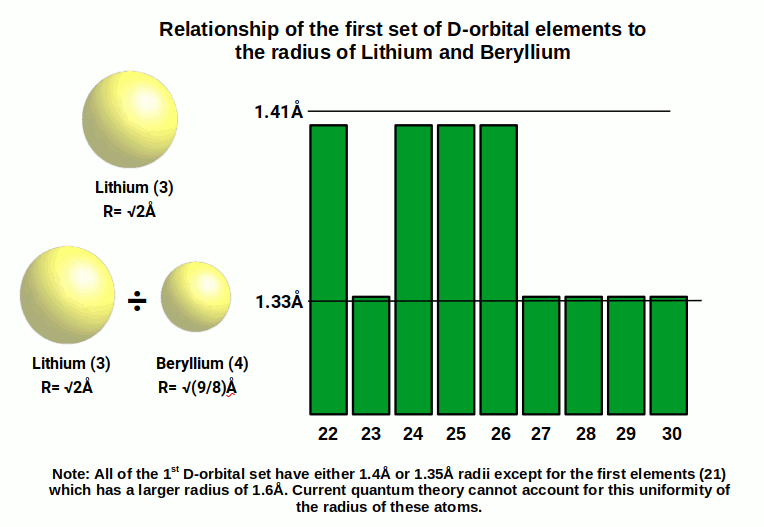
S-orbitals in the 5th shell
Once the 1st set of D-orbitals complete, up to zinc (30), another set of 6 P-orbitals form the 4th noble gas, Krypton (36). As the set forms, each of the subsequent radii gets smaller, up to the 3rd element of the set, Arsenic (33), whereby the radius stabilizes at 1.15Å. This value can be expressed as 2÷√3. A sphere of this radius will encompass an octahedron with a side length of √(8÷3).
If we apply the jitterbug transformation, we can expand the sphere into a cuboctahedron, and then add the corner parts to create a compound of the cube and octahedron. This is the same geometric process used previously to define the radius of Helium (2), and Lithium (3). This creates two new radii, one that surround the cube of 2Å, and a second that defined by the octahedron of 2.3Å. When we compare the results to the S-orbitals that from after the P-orbital set in the 5th Shell, rubidium (37) and strontium (38), we find a very close match.
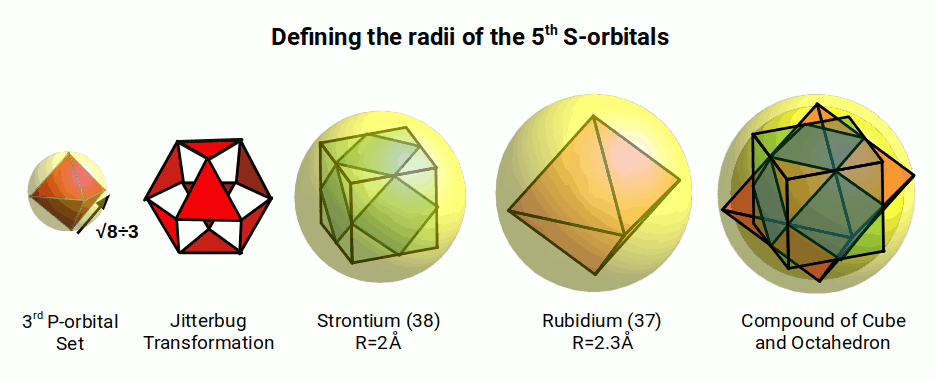
The experimentally determined value for Strontium (38) is 2Å, which provides an exact match to the geometric model. Rubidium (37) has an experimental radius of 2.35Å, which is 0.5Å larger than the geometric model, right on the cusp of the margin for experimental error. However, compared to the Bohr Model’s prediction of 2.65Å, and the Van der Wall radius of 3.08, the geometric interpretation is still much closer. The calculated values for Strontium (38) also overshoot the experimental values beyond the margin of error.
S-orbitals in the 6th shell
After Strontium (38) completes the 6th set of S-orbitals in the 5th shell, a 2nd set of D-orbitals forms in the 4th shell. These account for elements 39 to 48. After this, the 4th set 6 of P-orbitals elements form elements 49 to 54 in the 5th shell, culminating in the noble gas Xenon (54). As these P-orbital elements form, the subsequent radius of each is reduced in size to around 1.4Å, which is the same radius as Lithium (3). Then a final set of stable S-orbitals form.
Caesium (55) has a radius of 2.6Å, which is the largest of all elements on the periodic table. The final S-orbital element is Barium (56) with a radius of 2.15Å. Both of these radii can be found by multiplying 2÷√3 by √1.53, which results in a 2.12Å radius for Barium (56), and 2÷√3 by √1.54, to produce a 2.598Å for Caesium (55).
After the final S-orbital forms, the first set of F-orbital begin to appear in the 4th shell. Similar to the D-orbitals, these elements generally produce two specific radii of 1.85Å, falling to 1.75Å, which is close to √3, which can be determined by the multiplication of 2÷√3 by √1.5². This means that starting with the radius of the 3rd P-orbital set of 2÷√3Å, we can multiply by powers of √1.5 to find the orbital radii of the 4th P-orbitals (√2), and the F-orbitals (√3), and also the final stable S-orbital elements.

This relationship is driven by √1.5 which is the radius of the helium (2) atom. The other value of interest is 2÷√3, which is found when the Helium (2) radius is divided by the radius of Beryllium (4). This forms the basis for the other elements, that fall into multiples of the helium radius of √1.5.
This geometric model provides a much closer approximation to the S-orbital radii than the Bohr or Van der Waal radius. The Bohr model suggests a much larger radius of 2.98Å for Caesium, and 2.53Å for Barium, both of which are 0.38Å over the experimentally determined value. The Van der Waal radius is out even further, offering a radius of 3.43Å and 2.53Å for Caesium and Barium respectively. When we observe the complete set of S-orbital radii, it is clear that only the geometric ratio can provide an accurate description of each of the experimentally determined values.
When we look at the graph for all 12 S-orbital atoms, we can see that the Bohr radius does start to come close to predicting the Sodium and Magnesium radii. However, either side of this, the values are much less accurate, particularly for the first two atoms. The geometric model provides a consistently closer match to the experimental values than either the Bohr or Van der Waals predictions across the full set of S-orbital elements.
Conclusion
What does this tell us about S-orbitals?
This various radii of the 12 stable S-orbital elements are in Geometric ratio to each other. The first 4 atoms form the initial relationship, which is expanded through √1.5, √2, and √3 to define the orbital radii of all subsequent S-orbital Elements. This explains why the radius of each element does not expand in a quantized manner based on the increase in size of the atomic nucleus. The variation in the atomic radii is instead formulated around the geometric transformation of the electron cloud. The ratios found between the In, Out and mid-sphere of the cube, and the jitterbug transformation provide a much closer approximation to the experimentally verified atomic radii, than predictions provided by both the Bohr and Van der Waals models.
A new approach to the Electron cloud
Presently, no atomic theory is able to predict the radius of the experimentally determines radii of all Atoms. There is no explanation as to why hydrogen should be much smaller than helium, or why certain elements should be the same size, despite having vastly different numbers of protons and neutrons in their nucleus. Only the geometric model can offer an explanation, that is consistent for all stable elements. This makes it a more accurate predictor of atomic radii than either the Bohr or Van der Waals model across the full set of stable elements.




