Introduction
Every solid object you have ever touched — wood, metal, skin, stone — is made almost entirely of empty space. Atoms are so small that a single human hair is roughly one million atoms wide, yet each atom is itself 99.99% void, a tiny nucleus surrounded by electrons that occupy far more space than they take up. Understanding how scientists arrived at this picture is one of the great detective stories in the history of human thought.
This article traces that story from the first philosophical speculations in ancient India and Greece, through the experimental revolutions of the 19th and 20th centuries, to the quantum mechanical models used today — and beyond, to the geometric framework of Atomic Geometry that offers fresh insight into why atoms are shaped the way they are.
The concept of the atom has evolved over centuries, with ancient civilizations and notable scientists contributing to our understanding of its components and spatial configuration. Key milestones include the identification of electron shells, the realisation that electrons behave as both particles and waves, and the development of orbital models that can predict the structure of the periodic table. By studying the geometry and patterns of atoms, we can begin to unlock the secrets of natural processes and live in greater harmony with our environment.
Key takeaways
- Atoms are 99.99% empty space — the nucleus is vanishingly small relative to the electron cloud that surrounds it, a fact that overturned centuries of assumption about matter being solid.
- Each dominant atomic model — from Dalton's solid sphere to Bohr's planetary orbits to Schrödinger's wave functions — was displaced by a single decisive experiment or theoretical breakthrough, showing how radically our picture of the atom can shift.
- Atomic Geometry offers a geometric alternative to the Copenhagen interpretation, mapping S, P, D, and F orbitals onto three-dimensional forms and predicting the radii of all stable elements through spatial ratios rather than probability functions.
First notions of the atom
The journey begins in ancient India, where philosopher Acharya Kanada, in the 6th century BC, speculated that matter consisted of minute indivisible particles called the Iota. Kanada's observation laid the foundation for the concept of the atom. Fast-forward to ancient Greece, where philosophers Leucippus of Miletus and Democritus of Abdera coined the term "atomos" — meaning "uncuttable" — from which we derive the modern word "atom." Their ideas expanded on the notion put forth by the Indian philosophers.
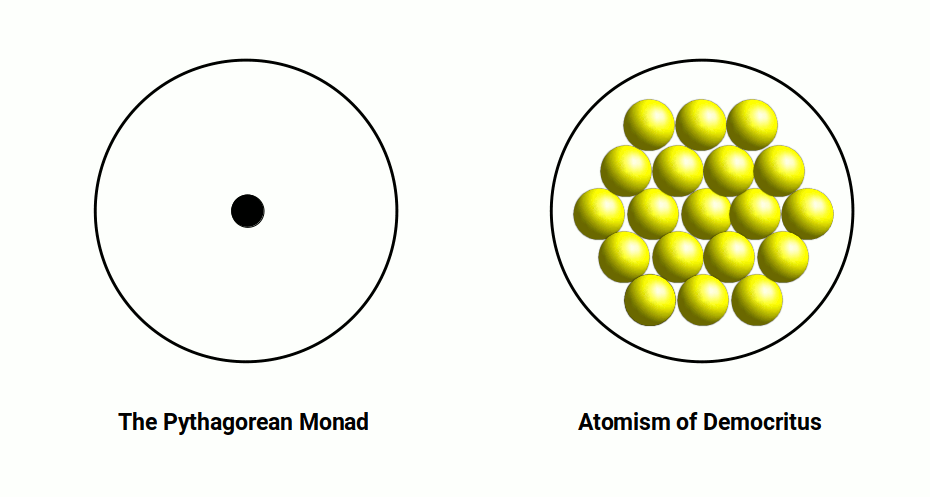
The exploration of the atom took a metaphysical turn within sacred geometry, with the introduction of the concept of the Monad, akin to a fundamental unit of existence. However, it was the discovery of the electromagnetic field that truly revolutionised scientific understanding. The realisation of this new force — capable of producing light and driving machines — prompted extensive experimental study throughout the 19th century.
First models of the atom
Chemist John Dalton, followed by J.J. Thomson, made significant contributions to the development of models representing the atom. Dalton's Solid Sphere model provided an initial understanding of the atom's structure, which was further expanded upon by Thomson's Plum Pudding model. This model depicted the atom as a spherical mass of positive matter with embedded electrons scattered throughout, like plums in a pudding.
Ernest Rutherford's famous gold foil experiment in 1911 shattered this picture. By observing alpha particles being deflected and reflected, Rutherford proposed the existence of the atomic nucleus, introducing the concepts of protons and a concentrated central charge. This discovery led to the acceptance that electrons orbit a central nucleus and occupy quantised energy shells.
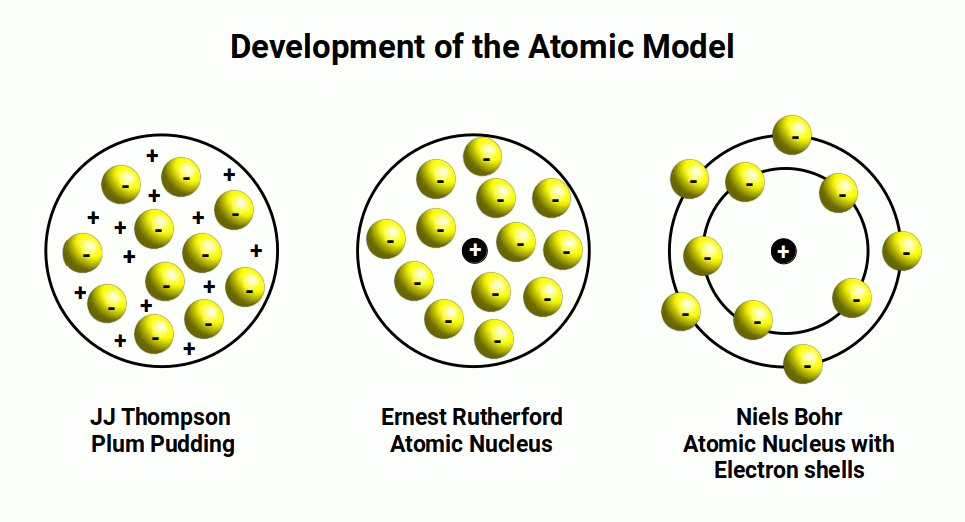
The mechanism by which electrons transition between shells, releasing quantised energy, puzzled scientists for some time. Albert Einstein's establishment of the photon as a quantised wave packet of energy, later formalised by Max Planck, provided crucial insights into this phenomenon. In 1913, Niels Bohr developed the Planetary model, which likened the atom's structure to our solar system — protons at the centre, electrons in fixed circular orbits. This model remains widely used in introductory chemistry to this day.
Wave-Particle Duality and the Electron
Whilst the Bohr model had revealed the quantised nature of the electron cloud, one major question remained unanswered: why did electrons not lose energy and collapse into the nucleus, as classical physics predicted they must? The answer came with the concept of the electron as a wave function. Louis de Broglie's Pilot Wave model proposed that every electron is guided by an associated matter wave, giving it a stable, non-radiating configuration. These developments challenged the prevailing belief that electrons were purely particles, revealing their wave-like properties — analogous in some ways to light. For a deeper exploration of this question, see our article on wave-particle duality.

Heisenberg's Uncertainty Principle, rooted in matrix mechanics, expanded upon these findings by proposing a probability function for electrons. This interpretation — known as the Copenhagen interpretation — posits that an electron exists in a superposition of all its possible states simultaneously until a measurement is made.
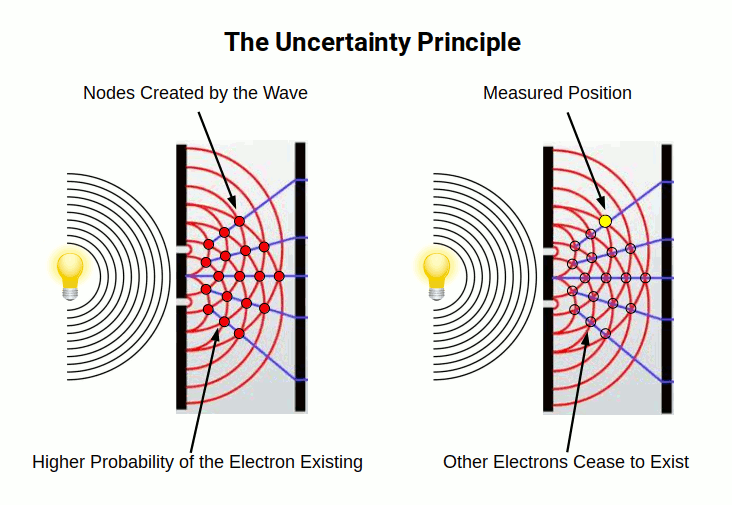
Wave equations
Erwin Schrödinger's equations in the 1920s collapsed the electron probability cloud into specific orbitals known as S, P, D, and F, giving a far richer picture of the atom's internal organisation. This interpretation, based on probability densities and quantised angular momentum, remains the foundation of modern atomic physics and chemistry. For a detailed look at the shapes these orbitals produce, see our articles on S orbital geometry and P orbital geometry.
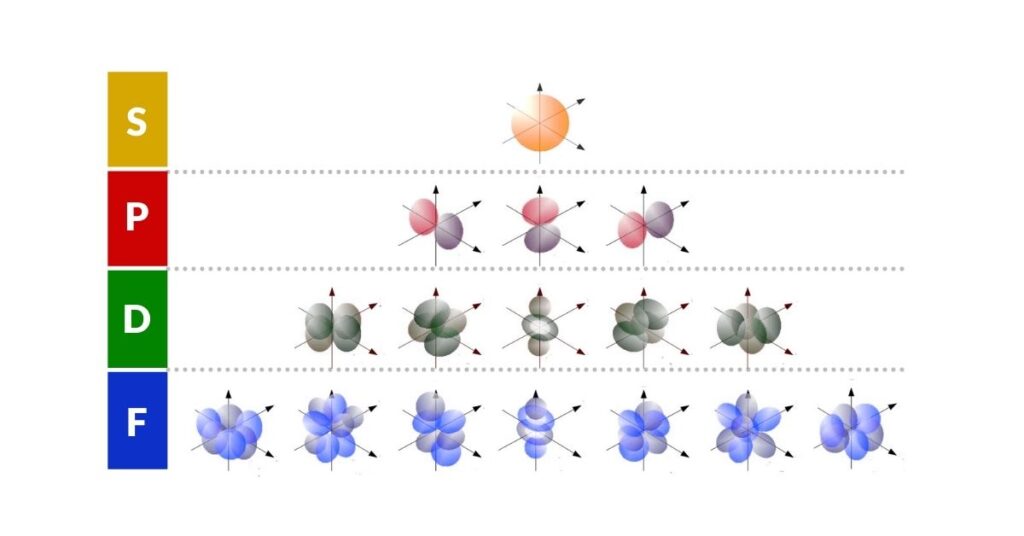
Throughout these discoveries, the atom's geometric nature became increasingly evident. While most of an atom is composed of empty space, comprehending the structure of that space allows a deeper understanding of the universe. You can also explore our article on the ultraviolet catastrophe and the Planck constant for the quantum story of how light and matter exchange energy.
Discovery of the neutron
In the 1930s, the discovery of the neutron through precise measurements added another crucial element to the atom's composition. This puzzle was solved by James Chadwick in 1932, who demonstrated that the nucleus contains electrically neutral particles of roughly the same mass as protons. This discovery completed the basic picture of the atom's nuclear structure and subsequently enabled the development of nuclear technology — with profound consequences during and after World War II.

Atomic Geometry
While the Copenhagen interpretation of the atom persists in mainstream academia, geometric perspectives offer a complementary lens through which to understand atomic structure. By mapping electron orbitals onto precise three-dimensional geometric forms, Atomic Geometry reveals patterns that align with the natural forms and ratios found throughout the physical world.
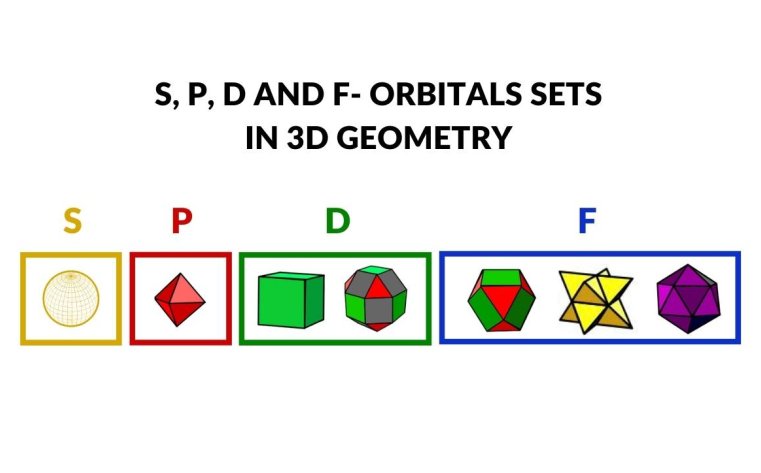
By delving into the atomic patterns of nature, we can unlock the insights necessary to harness nature's abilities — from splitting water for sustainable energy to living in greater harmony with our environment. The geometric model bridges the abstract mathematics of quantum mechanics with the tangible, visual language of form and space.
Conclusion
The concept of the atom has transformed throughout history, thanks to the contributions of ancient philosophers and pioneering scientists across 2,500 years. From the atomos of ancient Greece to the wave functions of quantum mechanics, our understanding has evolved from simple indivisible spheres to a rich landscape of orbitals, probability clouds, and geometric structure. As we continue to explore the geometric patterns and energetic potential of atoms, we move closer to a more profound understanding of our universe and our place within it.
FAQ
What did Rutherford discover in his gold foil experiment?
In 1911, Ernest Rutherford fired alpha particles at a thin sheet of gold foil. Most passed straight through, but a small number bounced back — something impossible if atoms were uniform spheres of matter. Rutherford concluded that nearly all of an atom's mass is concentrated in a tiny, dense, positively charged nucleus, with electrons orbiting at a relatively vast distance. This overturned Thomson's Plum Pudding model and introduced the nuclear atom.
Why did the Bohr model fail?
Niels Bohr's 1913 Planetary model explained why electrons occupy discrete energy levels, but it could not explain why electrons do not radiate energy and spiral into the nucleus, as classical electromagnetism predicts they should. It also broke down for atoms with more than one electron and could not account for the wave-like behaviour revealed by de Broglie and later Schrödinger. The model was eventually superseded by quantum mechanical orbital theory.
What is wave-particle duality in the context of the atom?
Wave-particle duality describes the experimental fact that electrons — and other quantum objects — behave as particles in some experiments and as waves in others. Louis de Broglie proposed in 1924 that every particle has an associated matter wave. Schrödinger's equations then described electrons not as point-like objects orbiting a nucleus but as standing wave functions spread across specific orbitals. Heisenberg's Uncertainty Principle formalised the limits of knowing both position and momentum simultaneously.
What is Atomic Geometry?
Atomic Geometry is a geometric model of the atom that we developed, which maps the S, P, D, and F electron orbitals onto precise three-dimensional geometric forms. Unlike the statistical Copenhagen interpretation, Atomic Geometry uses spatial ratios to predict the radii of all stable elements in the periodic table. It offers a visually intuitive framework that connects atomic structure to the broader geometric patterns found throughout nature.




