Introduction
The periodic table contains one of chemistry's most persistent anomalies: elements 43 (technetium) and 61 (promethium) are the only two elements with atomic numbers below 83 that have no stable isotopes whatsoever. Every other element in that range — all 79 of them — has at least one stable form. These two do not. The Standard Model attributes this to unfavourable proton-to-neutron ratios, but provides no explanation for why the pattern breaks at exactly these two positions and nowhere else in the sequence.
Harmonic Chemistry proposes a precise answer: the positions of elements 43 and 61 are predicted by the same interval mathematics that structures the musical scale. The argument rests on three numbers — 84, 42, and 17 — and requires no specialist knowledge of either music or chemistry to follow.
Key Takeaways
- Elements 43 and 61 are the only unstable elements below bismuth (83) — their positions are predicted by musical interval ratios
- 84 = 12 × 7 — the product of the 12 chromatic notes and 7 natural tones per octave — defines the boundary of stable chemistry
- The midpoint at 42, the third at 28, and the fifth at 56 each correspond to real structural boundaries in atomic physics
- The 17 stable elements between 43 and 61 match the 17-note Arabic scale; the 22 stable elements after 61 match the 22-note Vedic Shruti system
- Harmonic Chemistry complements Atomic Geometry — geometry defines orbital shape; resonance defines stability
The Numbers First
Before any music theory or chemistry, the core argument can be stated in pure arithmetic.
There are 84 elements from hydrogen (1) to polonium (84). Beyond polonium, all elements are radioactive. The number 84 divides cleanly as follows:
- 84 ÷ 2 = 42 — the midpoint of the sequence. Element 42 is molybdenum, the last stable element before the first anomaly. Element 43 is the break.
- 84 ÷ 3 = 28 — one third of the sequence. Element 28 is nickel.
- 28 × 2 = 56 — two thirds of the sequence. Element 56 is barium, which marks a real boundary in atomic structure.
- 44 to 60 = 17 elements — the count of stable elements between the two anomalies.
- 62 to 83 = 22 elements — the count of stable elements from the second anomaly to the end of the stable sequence.
The numbers 12, 7, 17, and 22 are the fundamental quantities of musical scale theory. The number 84 = 12 × 7 connects them to the periodic table — and 42 is simply 84 divided by 2. That parallel is what Harmonic Chemistry proposes to explain.
Why 84? The Musical Scale
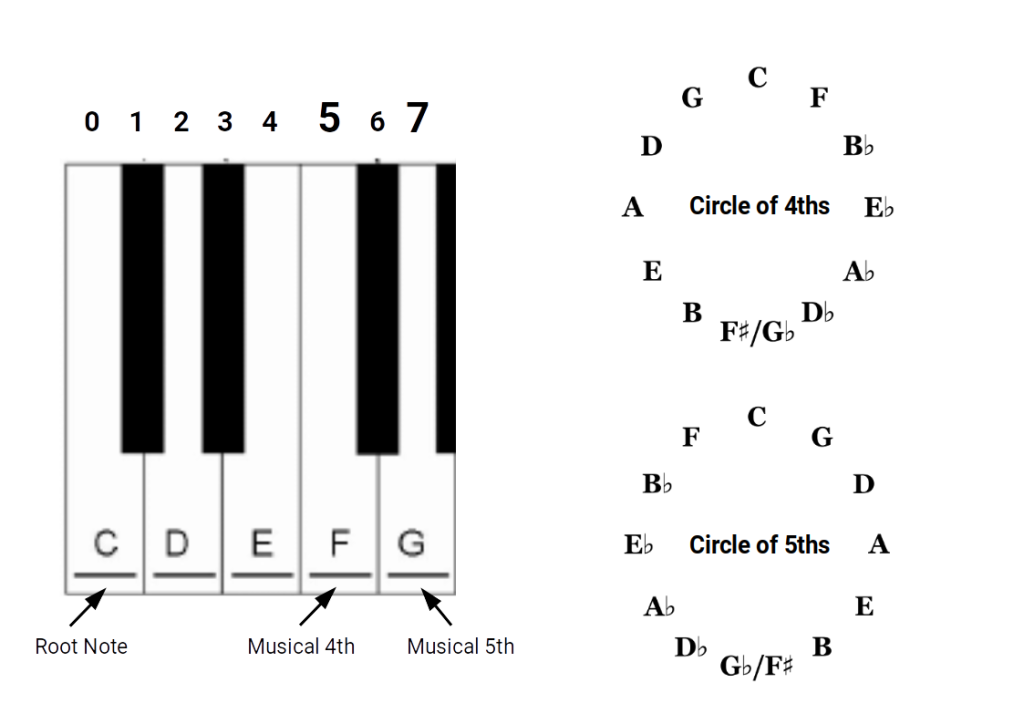
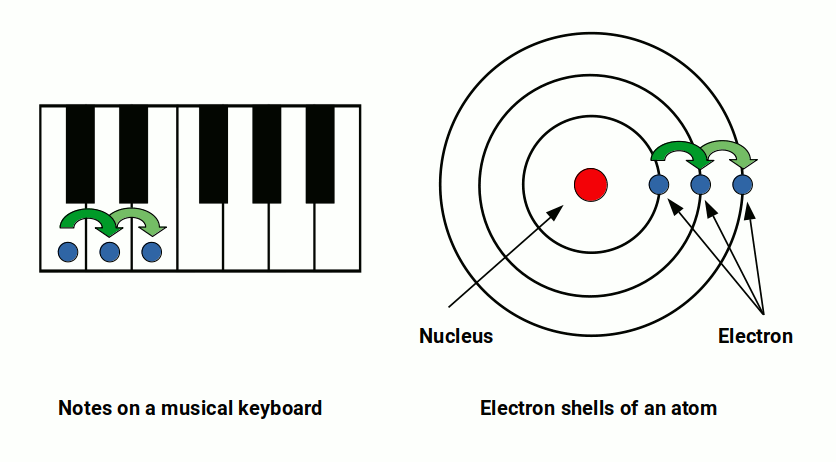
The 12-note chromatic scale — the foundation of Western music — is built from two intervals: the perfect fourth and the perfect fifth. These are derived from the simplest possible string divisions: dividing a string into 3 produces the fifth; dividing into 4 produces the fourth. From these two ratios, applied repeatedly, all 12 chromatic notes are generated.
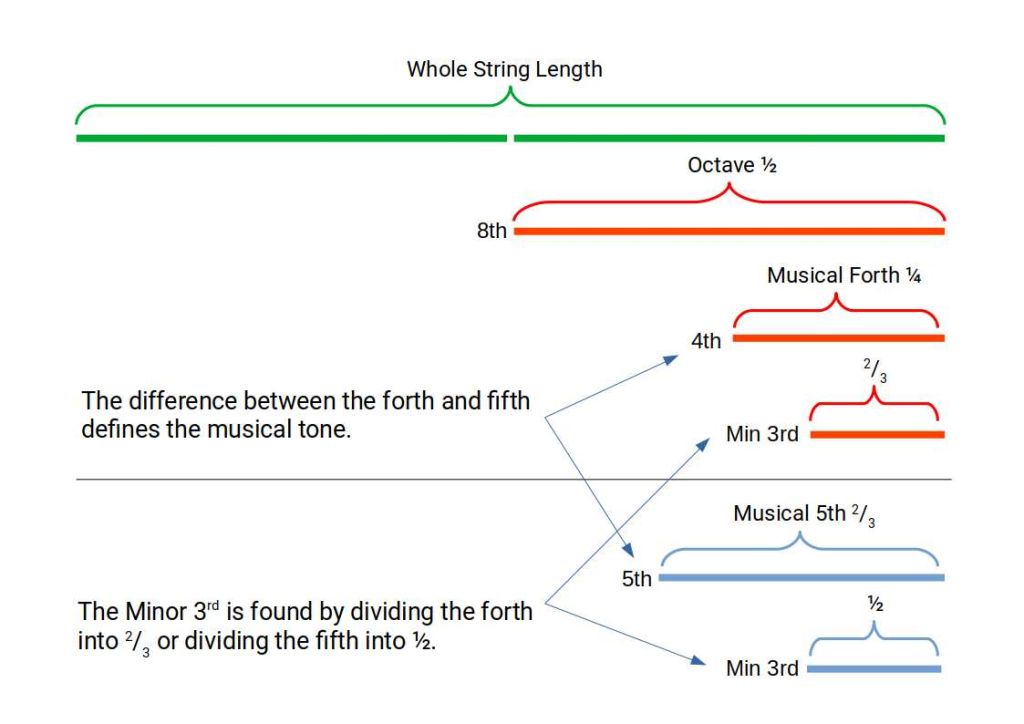
The resulting scale has 12 notes per octave: 7 natural tones (the white keys on a piano) and 5 accidentals (the black keys). The product 12 × 7 = 84. This is the same 84 that defines the boundary of stable chemistry. Harmonic Chemistry proposes this is not a coincidence — the periodic table and the musical scale are both expressions of the same underlying resonance structure.

The Break at 42: The Octave
In music, the octave is the most fundamental division: the point at which a frequency doubles and the pattern restarts. The 12-note chromatic scale divides symmetrically — 6 notes on each side of the midpoint, with the tritone sitting at the exact centre and the octave completing the cycle at position 12.
The midpoint of the 84-element sequence is 42. Element 42 is molybdenum — the last stable element before the sequence breaks. Element 43 (technetium) is the first element with no stable isotopes.
The musical name for this position is the octave. The atomic consequence is instability. Both occur at exactly the same arithmetic position — the midpoint of a 12 × 7 sequence.
The Fifth at 56: The Aufbau Inversion
Dividing 84 by 3 gives 28; doubling that gives 56 — two thirds of the way through the sequence. In music, this position is the perfect fifth: the second most fundamental interval after the octave, and the first interval generated by dividing a string into three.
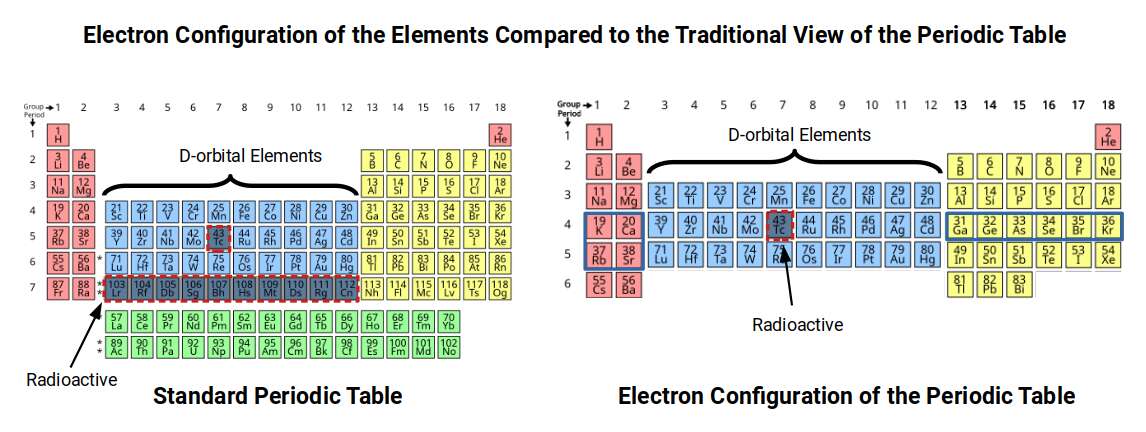
Element 56 (barium) marks a genuine boundary in atomic physics. It is the last element whose outermost electron fills an S-orbital in the 6th shell. From element 57 onward, the filling sequence inverts: the F-orbitals (elements 57–70) fill in the 4th shell, and the final D-orbitals (elements 71–80) fill in the 5th shell — lower shells than the preceding S-orbital. This inversion is called the Aufbau anomaly, and element 56 is its threshold.
The musical fifth and the Aufbau inversion boundary fall at the same position: 56, two thirds of 84.
The Reordered Periodic Table
When the periodic table is redrawn with each orbital type placed in its true shell position — rather than its conventional row — a striking symmetry emerges.
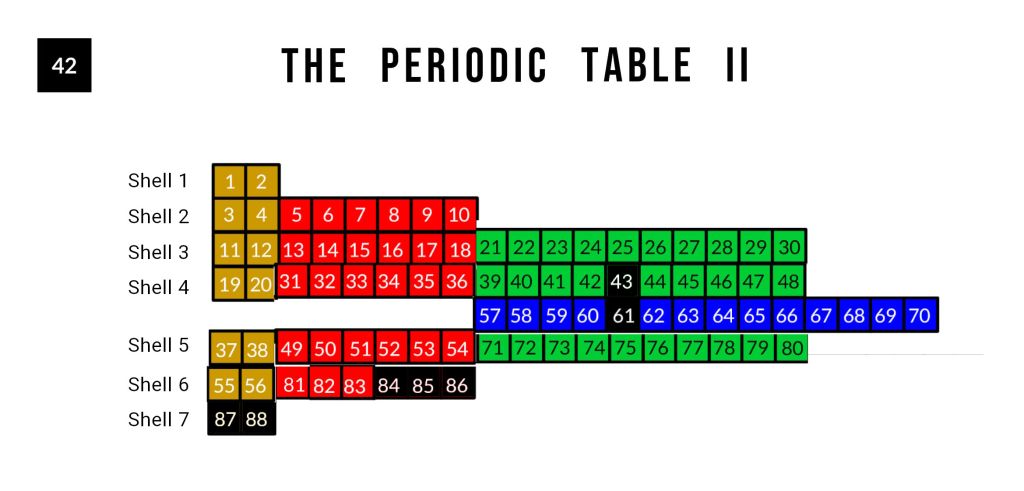
Elements 43 and 61 occupy the same relative position within their orbital blocks. In the conventional periodic table, this symmetry is hidden by the layout. In the reordered view, it is immediately apparent. Their instability is not a pair of unrelated anomalies — it is the same geometric break occurring twice, at the octave and at the Arabic scale boundary, in successive segments of the sequence.
The Arabic Scale: 17 Elements Between the Anomalies
After element 43, there are exactly 17 stable elements before the next anomaly — elements 44 through 60 inclusive.
The number 17 is not arbitrary in music. In the 13th century, the Persian-Iraqi theorist Safi al-Din Urmawi developed a 17-tone scale to describe Arabic and Persian music, later formalised by Alexander J. Ellis. It is built from the same perfect fourths and fifths that generate the Western chromatic scale — but it treats sharps and flats as distinct tones rather than enharmonic equivalents, producing 17 separate pitches per octave: 7 naturals, 5 sharps, and 5 flats.
C, D♭, C♯, D, E♭, D♯, E, F, G♭, F♯, G, A♭, G♯, A, B♭, A♯, B, (C)
The final C appears in brackets as the octave completion — the same note as the first, one frequency doubling higher. The 17 distinct tones are the notes from C to B♮. The count of stable elements between the two anomalies is exactly 17. The count of tones in the Arabic scale is exactly 17. Harmonic Chemistry proposes these are the same number for the same reason.
The Vedic Shruti Scale: 22 Elements After the Second Anomaly
After element 61, there are exactly 22 stable elements before the end of the stable sequence — elements 62 through 83 inclusive.
The Vedic system of Shruti divides the octave into 22 intervals — described as the smallest pitch increments the human ear can reliably distinguish. These 22 Shruti subdivide the 7 principal tones (Swara) of the Indian classical scale into their finest gradations.
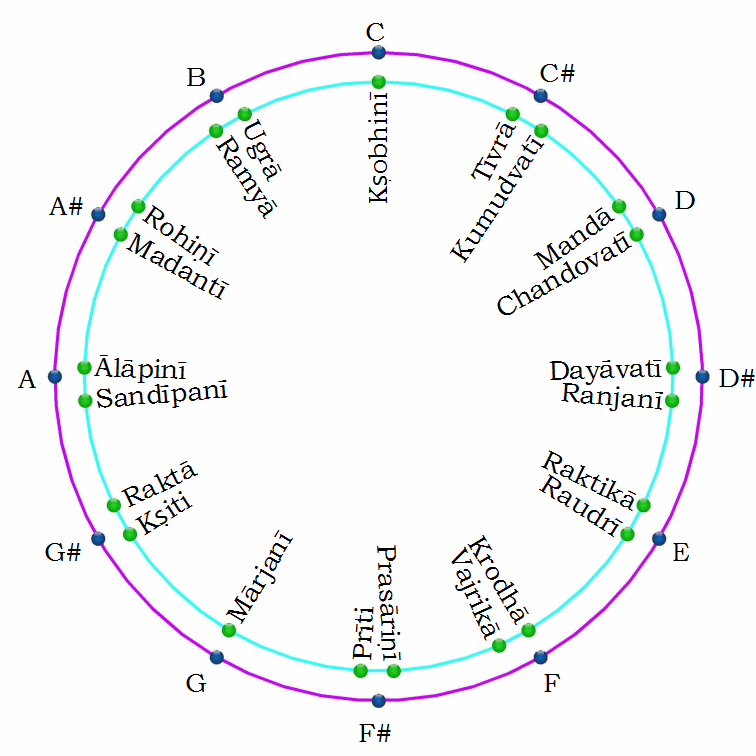
Within the Shruti system, only two of the 22 positions are single-valued: the root (the starting note) and the perfect fifth. Every other position has two variants — a sharp and a flat version. The perfect fifth in this context corresponds to the musical fifth boundary at element 56: the single structural landmark within the sequence that does not shift between orbital shells, just as the Shruti fifth does not split into two variants.
The count of stable elements from 62 to 83 is exactly 22. The count of Shruti in the Vedic octave is exactly 22.
Conclusion
Three exact numerical correspondences — 17, 22, and the midpoint at 42 — connect the positions of the only two unstable elements below bismuth to the structure of the musical scale. None of these matches is approximate: 17 stable elements between the anomalies, 22 stable elements after the second, and the first anomaly at position 43 = 42 + 1, the note immediately after the octave midpoint.
The number 84 = 12 × 7 encodes both the musical scale and the boundary of stable chemistry. The divisions at 28 (a third), 42 (the octave midpoint), and 56 (the fifth) correspond to real structural boundaries in orbital shell physics. Harmonic Chemistry does not replace the quantum mechanical description of the atom — it reveals the resonant structure within which that description operates, and connects it to Atomic Geometry and Geo-Quantum Mechanics as a unified geometric account of the periodic table.
FAQ
What is Harmonic Chemistry?
Harmonic Chemistry is a model that maps the structure of the periodic table onto the mathematics of musical scales. By treating the 84-element sequence from hydrogen to polonium as analogous to the notes of a musical keyboard — 12 notes, 7 white and 5 black per octave — it provides a geometric explanation for why elements 43 and 61 are the only two unstable atoms in an otherwise continuous sequence of stable elements.
Why are elements 43 and 61 unstable?
In Harmonic Chemistry, the 84-element sequence divides at its midpoint — 42 — exactly as a musical octave divides a scale at its midpoint. Element 43 falls at this break. Element 61 falls at the boundary of the 17-note Arabic scale segment (elements 44–60), where the next resonant interval begins. Both positions are predicted by the same interval mathematics that structures the chromatic musical scale.
What is the connection between the periodic table and musical intervals?
The number 84 = 12 × 7 — the product of the 12 chromatic notes and the 7 natural tones per octave. Dividing 84 by 2 gives 42 (the octave midpoint, predicting element 43). Dividing by 3 gives 28 (the musical third); doubling gives 56 (the musical fifth). Element 56 marks a real atomic boundary — the last S-orbital in the 6th shell before the Aufbau filling sequence inverts.
What is the significance of the Arabic and Vedic scales?
After element 43, there are exactly 17 stable elements before the next unstable one (elements 44–60). The 17-note Arabic scale — developed by Safi al-Din Urmawi in the 13th century — contains exactly 17 tones per octave. After element 61, there are exactly 22 stable elements before bismuth (83). The Vedic Shruti scale contains exactly 22 tones — the smallest intervals the ear can discern. Both counts are exact, not approximate.
How does Harmonic Chemistry relate to Atomic Geometry?
Atomic Geometry defines the geometric shapes of the S, P, D, and F orbitals. Harmonic Chemistry uses musical ratios to explain why specific positions within those orbital types are stable or unstable, and why the Aufbau filling sequence deviates at certain atomic numbers. Geometry defines the shape of each orbital; resonance defines which positions within those orbitals are stable.

