Introduction
Molecular geometry — the study of the three-dimensional shapes of molecules — is a foundation of modern chemistry. It explains bond angles, polarity, and reactivity. But it stops at three dimensions. Many of the most important phenomena in chemistry and biology — electromagnetic conductivity in crystal lattices, the anomalous ease of nitrogen fixation by bacteria, the geometric regularity of the five biogeochemical cycles — remain incompletely explained by 3D molecular geometry alone.
Geo-Molecular Chemistry extends this framework into four dimensions. By applying the geometric transformations of Atomic Geometry and Geo-Quantum Mechanics to molecular systems, it provides geometric explanations for properties that emerge from the lattice structure of matter rather than from individual atomic bonds.
Key Takeaways
- Geo-Molecular Chemistry extends molecular geometry into 4D — applying polyhedral transformations to the atomic lattice
- The Extended Jitterbug transformation explains how lattice geometry produces electromagnetic conductivity, resistance, and thermal properties
- All five molecules central to the biogeochemical cycles share the same tetrahedral geometry — suggesting a unified geometric basis for the chemistry of life
- Nitrogen fixation by bacteria — one of biology's great unsolved mechanisms — is proposed as a geometric transformation from diatomic (linear) to tetrahedral molecular structure
- The five biogeochemical cycles map onto the five Platonic solids — connecting planetary chemistry to the same polyhedral framework that governs atomic structure
Molecular Geometry and the Atomic Lattice
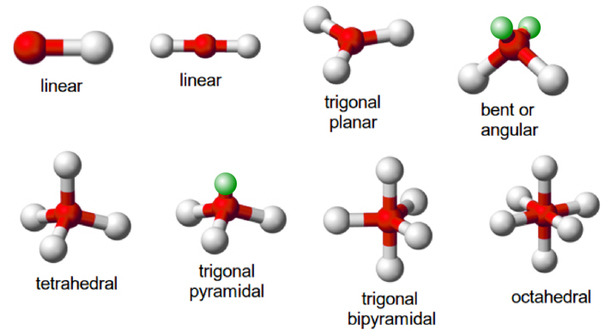
Standard molecular geometry describes the shape of individual molecules: water is bent, methane is tetrahedral, nitrogen gas is linear. These shapes determine how molecules interact, react, and bond. The standard model is powerful for isolated molecules but has limits when applied to extended lattice structures — the repeating geometric arrays that make up crystals, metals, and biological macromolecules.
In an atomic lattice, the arrangement of atoms is not static — it shifts in response to temperature, pressure, and electromagnetic fields. These shifts are geometric transformations, and Geo-Molecular Chemistry proposes that the specific transformations available to a given lattice — determined by the polyhedra that describe its atomic geometry — directly produce its observable physical properties.
The Extended Jitterbug in the Lattice
The key geometric transformation in Geo-Molecular Chemistry is the Extended Jitterbug — the continuous transformation connecting five polyhedra: Octahedron, Icosahedron, Cuboctahedron, Snub Cube, and Rhombic-Cuboctahedron. This transformation, introduced in Geo-Quantum Mechanics, governs the radii of electron shells across the periodic table. At the molecular scale, it also governs how atomic lattices shift between configurations.
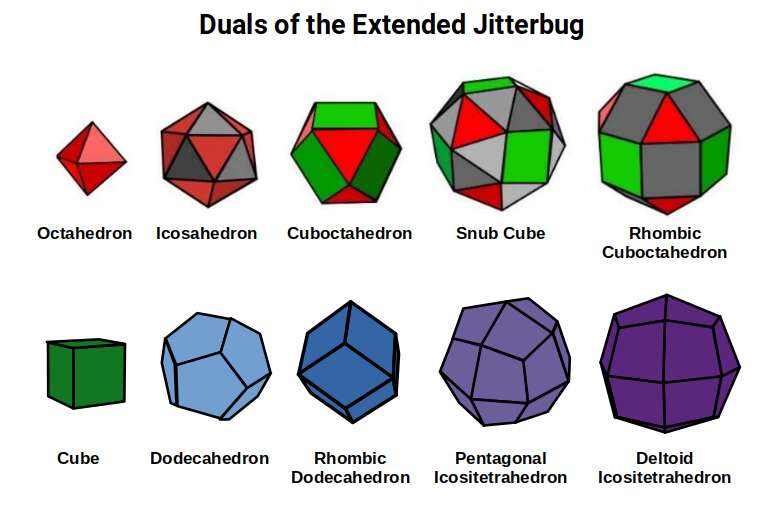
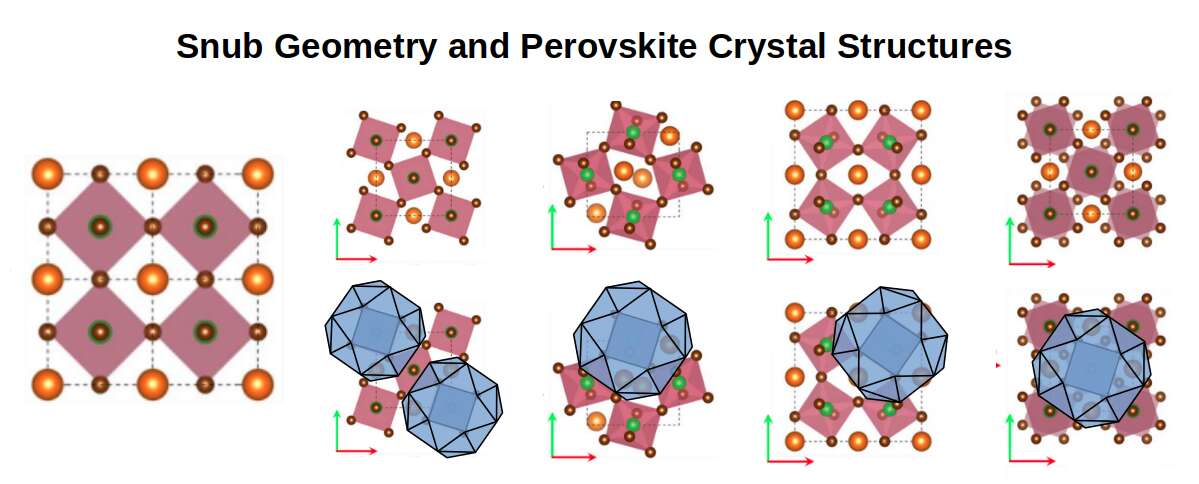
When a lattice undergoes the Jitterbug transformation — shifting from one polyhedral configuration to another — its electron pathways change. A lattice in the Cuboctahedral configuration allows electrons to flow freely in multiple directions, producing high conductivity. A lattice that shifts toward the Octahedral configuration tightens its geometry, constraining electron flow and increasing resistance. Thermal energy drives these transitions, connecting the geometric model directly to observed temperature-dependent conductivity. Perovskites — whose lattice geometry corresponds to the Snub Cube position in the Extended Jitterbug sequence — exemplify this relationship: their extraordinary electronic properties, which make them candidates for next-generation solar cells and superconductors, are consistent with the high geometric symmetry of that lattice configuration.
The Five Biogeochemical Cycles
Life on Earth is sustained by five biogeochemical cycles: the water cycle, the carbon cycle, the nitrogen cycle, the sulphur cycle, and the phosphorus cycle. Each cycle moves a key element or compound through the atmosphere, biosphere, and geosphere in a continuous loop. Together they regulate the conditions that make complex life possible.
What is remarkable about these five cycles is the geometry of the molecules at their core:
- Water cycle — water (H₂O) appears bent when viewed as atoms alone, but its underlying electron geometry is tetrahedral: four electron pairs around the oxygen atom — two bonding, two lone — arranged at tetrahedral angles. The bent molecular shape is simply what a tetrahedron looks like when two of its vertices are occupied by invisible lone pairs rather than atoms
- Carbon cycle — methane (CH₄) is a perfect tetrahedron: one carbon atom at the centre, four hydrogen atoms at each vertex
- Nitrogen cycle — ammonia (NH₃) is a trigonal pyramidal molecule derived from a tetrahedron with one lone pair replacing a hydrogen
- Sulphur cycle — sulphate (SO₄²⁻) is a perfect tetrahedron with an oxygen atom at each vertex
- Phosphorus cycle — phosphate (PO₄³⁻) is likewise tetrahedral, structurally identical to sulphate
All five key molecules share tetrahedral geometry. Geo-Molecular Chemistry proposes this is not coincidental — the tetrahedron is the simplest three-dimensional form, and life is organised around it because tetrahedral geometry provides the most stable platform for the chemical bonds that transfer energy through biological systems.
Diatomic Molecules and the Geometry of Air
Before molecules enter the biogeochemical cycles, they must first transition from their elemental diatomic forms. The air we breathe is composed almost entirely of diatomic molecules: nitrogen (N₂), oxygen (O₂), and smaller quantities of others. A diatomic molecule is geometrically the simplest possible form — two atoms connected by a single bond axis, a line.
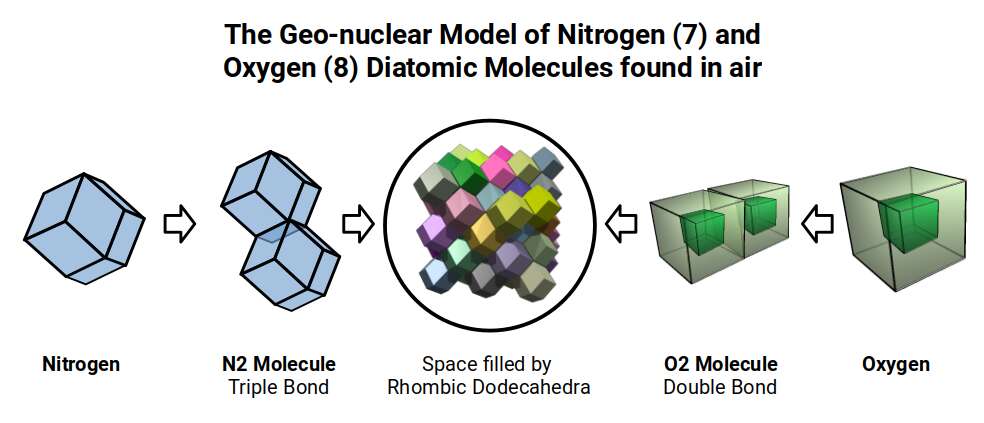
The transition from a diatomic line to a tetrahedral molecule — from one dimension to three — is the geometric event at the heart of biochemistry. It is the event that Geo-Molecular Chemistry proposes as the key to understanding nitrogen fixation.
The Nitrogen Fixation Problem
The nitrogen cycle depends on a process called nitrogen fixation — the conversion of diatomic nitrogen gas (N≡N) into ammonia (NH₃) or other bioavailable nitrogen compounds. This is essential for the synthesis of proteins and nucleic acids. Without it, life as we know it would not exist.
The triple bond in N≡N is one of the strongest in chemistry — requiring approximately 945 kJ/mol to break. Industrial nitrogen fixation (the Haber-Bosch process) requires temperatures of 400–500°C and pressures of 150–300 atmospheres. Yet certain bacteria accomplish the same transformation under ambient conditions — at room temperature, normal atmospheric pressure, using an enzyme called nitrogenase. How they do this remains, in the words of biochemists, one of the most important unsolved problems in biology.
Nitrogenase does consume energy — approximately 16 ATP molecules per nitrogen fixed — but this is far less than the energy equivalent required by the Haber-Bosch process operating at extreme temperature and pressure. The question is not whether energy is consumed, but why the same geometric transformation is achievable under ambient conditions by an enzyme when industry requires a sledgehammer.
Geo-Molecular Chemistry proposes that the nitrogenase enzyme creates a geometric cavity whose 4D field geometry induces the transition from linear to tetrahedral structure — enabling the nitrogen atoms to reorganise with the available ATP input rather than requiring brute-force bond breaking. The geometry does the work that temperature and pressure do industrially — consistent with the vacuum geometry described in 4D Aether Theory, where the structure of space itself carries energy that can be accessed through the right geometric configuration.
Geometric Biology: Haemoglobin and Chlorophyll
The geometric approach extends to the large biological molecules that power animal and plant life.
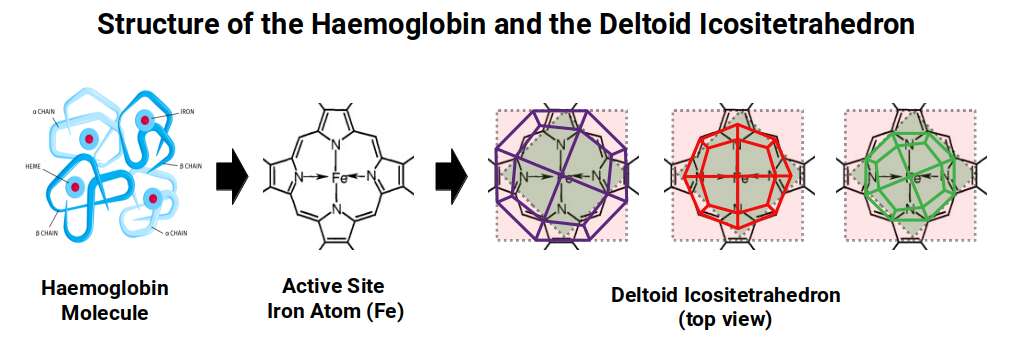
Haemoglobin — the protein that carries oxygen in red blood cells — has four iron-containing haem groups arranged in a roughly tetrahedral configuration. The cooperative binding of oxygen (where binding at one site increases affinity at others) is a consequence of this four-subunit geometric arrangement. Geo-Molecular Chemistry maps the haemoglobin structure to the Deltoid Icositetrahedron, the dual of the Snub Cube — a 24-faced polyhedron whose four-subunit organisation closely mirrors the tetrahedral arrangement of haemoglobin's haem groups, connecting the most important protein in animal biology to the Extended Jitterbug sequence.
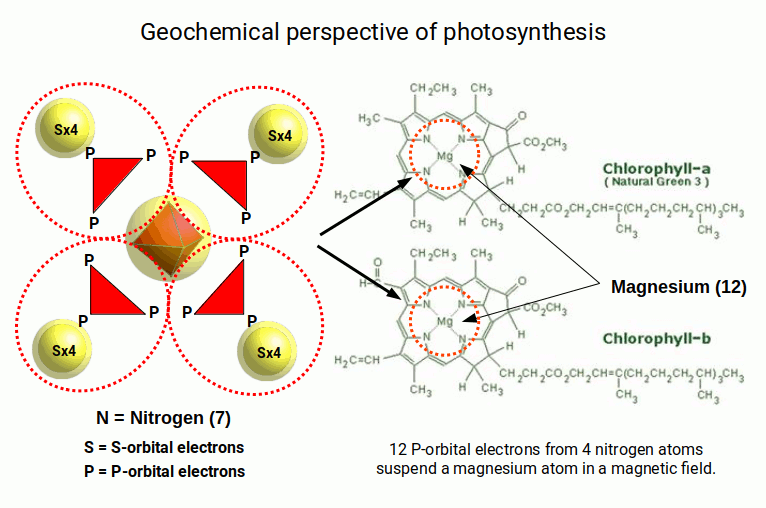
Chlorophyll — the molecule that captures sunlight in plants — centres on a magnesium atom held in a planar square coordination by a porphyrin ring. This square geometry corresponds directly to the D-orbital geometry identified in Atomic Geometry. The specific wavelengths of light absorbed by chlorophyll (red ~680 nm and blue ~450 nm, with green reflected) are a consequence of the energy levels defined by this square coordination geometry.
Conclusion
Geo-Molecular Chemistry proposes that the geometric transformations governing atomic structure — the Extended Jitterbug, tetrahedral symmetry, and 4D lattice transitions — also govern the behaviour of molecules, crystals, and biological systems. The tetrahedral geometry shared by all five biogeochemical cycle molecules is not accidental: it is the simplest stable three-dimensional structure, and life is built on it. The mystery of nitrogen fixation resolves as a 4D geometric transition. The cooperative oxygen binding of haemoglobin and the light absorption of chlorophyll follow from the same polyhedral principles that structure the periodic table.
Together with Harmonic Chemistry and Atomic Geometry, Geo-Molecular Chemistry forms part of a unified geometric account of chemistry — from the nucleus outward to the molecules of life.
FAQ
What is Geo-Molecular Chemistry?
Geo-Molecular Chemistry extends conventional molecular geometry into four dimensions. Where standard molecular geometry describes the three-dimensional shape of molecules, Geo-Molecular Chemistry examines how geometric transformations of the atomic lattice — particularly the Extended Jitterbug — give rise to physical properties such as electromagnetic conductivity, resistance, and thermal behaviour.
What is the Extended Jitterbug and why does it matter for chemistry?
The Extended Jitterbug is a continuous geometric transformation connecting five polyhedra — the Octahedron, Icosahedron, Cuboctahedron, Snub Cube, and Rhombic-Cuboctahedron. In Geo-Molecular Chemistry, this transformation describes how atomic lattice structures shift between configurations, producing measurable changes in electrical conductivity, magnetic order, and thermal properties.
What are the five biogeochemical cycles and how does geometry explain them?
The five biogeochemical cycles — water, carbon, nitrogen, sulphur, and phosphorus — are the biochemical systems that power all life on Earth. Geo-Molecular Chemistry maps each cycle to a nested set of five Platonic solids. Each molecule central to a given cycle — water (tetrahedral), methane (tetrahedral), ammonia (tetrahedral), sulphate (tetrahedral), phosphate (tetrahedral) — shares the same underlying tetrahedral geometry, suggesting a common geometric foundation for the chemistry of life.
How does Geo-Molecular Chemistry explain nitrogen fixation?
Nitrogen fixation — the breaking of the triple bond in diatomic nitrogen (N≡N) by bacteria — is one of the most energy-intensive chemical processes in biology, yet bacteria achieve it under ambient conditions with no apparent external energy source. Geo-Molecular Chemistry proposes that the geometric transformation from a diatomic molecule (a line) to a tetrahedral molecule is the underlying mechanism — the same 4D geometric transition that drives the Extended Jitterbug transformation.
How does Geo-Molecular Chemistry relate to Geo-Biology?
Geo-Biology applies the geometric framework of Geo-Molecular Chemistry to living systems — examining how the five biogeochemical cycles interact geometrically to sustain the biosphere. Where Geo-Molecular Chemistry focuses on individual molecules and lattice transformations, Geo-Biology scales the same geometric principles to ecosystems and the planetary cycling of matter.

